Abstract
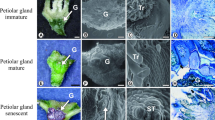
This study compares oil and mucilage idioblasts occurring together in the vegetative organs of Ocotea pulchella, a Lauraceae species. Our focus is specifically on the ontogeny and developmental cytology of these secretory cells. Both types of idioblasts originate from solitary cells located in the fundamental meristem, underlying the protodermis. The growth of both types of idioblasts is asynchronous, with the oil idioblasts developing first, but their initiation is restricted to the early stages of organ development. Mucilaginous idioblasts occur exclusively in the palisade parenchyma, while oil idioblasts are scattered throughout the mesophyll, midrib, and petiole of the leaves. The lamellar secretion of mucilage idioblasts is mostly made up of polysaccharides, while the secretion of oil idioblasts is made up of terpenes and lipids. Cupule occurred only in the oil idioblasts, while suberized layers occurred in both types of cells. We found that immature oil idioblasts that are close to each other fuse; mature mucilage idioblasts have labyrinthine walls arranged in a reticulate pattern; the cells close to the oil idioblasts have a pectin protective layer; and the oil idioblasts have a sheath of phenolic cells. In contrast to previous reports, the two types of secretory idioblasts were recognized during the early stages of their development. The results emphasize the importance of combining optical and electron microscopy methods to observe the ontogenetic, histochemical and ultrastructural changes that occur during the development of the secretory idioblasts. This can help us understand how secreting cells store their secretions and how their walls become specialized.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Baas P, Gregory M (1985) A survey of oil cells in the dicotyledons with comments on their replacement by and joint occurrence with mucilage cells. Isr J Plant Sci 34:167–186. https://doi.org/10.1080/0021213X.1985.10677020
Baitello JB, Marcovino JR (2003) Lauraceae. Ocotea Aubl. In: Wanderley et al (ed) Flora Fanerogâmica do Estado de São Paulo, vol. 3. Ed. RiMa, São Carlos, p 186
Bakker ME, Baas P (1993) Cells walls and mucilage cells. Acta Bot Neerl 42:133–139
Bakker ME, Gerritsen AF (1989) A suberized layer in the cell wall of mucilage cells of Cinnamomum. Ann Bot 63:441–448. https://doi.org/10.1093/oxfordjournals.aob.a087764
Bakker ME, Gerritsen AF (1990) Ultrastructure and development of oil idioblasts in Annona muricata L. Ann Bot 66:673–686. https://doi.org/10.1093/oxfordjournals.aob.a088082
Bakker ME, Gerritsen AF (1992) Oil and mucilage cells in Annona (Annonaceae) and their systematic significance. Blumea: biodiversity. Evol Biogeogr Plants 36:411–438
Bakker ME, Gerritsen AF, Van Der Schaaf PJ (1991) Development of oil and mucilage cells in Cinnamomum burmanni. An ultrastructural study. Acta Bot Neerl 40(339):356. https://doi.org/10.1111/j.1438-8677.1991.tb01564.x
Bannister JM, Conran JG, Lee DE (2012) Lauraceae from rainforest surrounding an early Miocene maar lake, Otago, southern New Zealand. Rev Palaeobot Palynol 178:13–34
Barnett JR, Cooper P, Bonner L (1993) The protective layer as an extension of the apoplast. IAWA J 14:163–171
Betim FCM, Barbosa V, de Oliveira CF, Miguel OG, Raman V, Budel JM, Miguel MD, Dias JDFG (2020) Microscopy and histochemistry of Ocotea nutans (Nees) Mez (Lauraceae). Flora 273:151708. https://doi.org/10.1016/j.flora.2020.151708
Brustulim LJR, Monteiro LM, Almeida VPD, Raman V, Maia BHLDNS, Casapula I, Paludo KS, Bussade JE, Rehman JUR, Kahn IA, Farrago PV, Budel, JM (2020) Ocotea porosa: anatomy and histochemistry of leaves and stems, chemical composition, cytotoxicity and insecticidal activities of essential oil. Braz Arch Biol Technol 63. https://doi.org/10.1590/1678-4324-2020190082
Cain AJ (1947) The use of Nile blue in the examination of lipoids. J Cell Sci 3:383–392. https://doi.org/10.1242/jcs.s3-88.3.383
Canaveze Y, Mastroberti AA, Mariath JEA, Machado SR (2019) Cytological differentiation and cell wall involvement in the growth mechanisms of articulated laticifers in Tabernaemontana catharinensis A.DC. (Apocynaceae). Protoplasma 256:131–146. https://doi.org/10.1007/s00709-018-1284-3
Carvalho PER (2006) Canela-do-brejo: Ocotea pulchella. In: Espécies Arbóreas Brasileiras, vol 2. Brasília, DF: Embrapa Informação Tecnológica; Colombo: Embrapa Florestas, pp 133–140
Castro MM, Demarco D (2008) Phenolic compounds produced by secretory structures in plants: a brief review. Nat Prod Commun 3(8):1273–1284. https://doi.org/10.1177/1934578X0800300809
Chaffe SC (1974) Cell formation and protective layer development in the xylem parenchyma of trembling aspen. Protoplasma 89:335–354
Chu QG, Liu WZ, Hu ZH (1999) Comparative studies on the distribution and structure of oil cells and mucilage cells in the leaves of 13 species of Lauraceae. Chin J Appl Environ Biol 5:165–169
Chu QG, Hu ZH (1999) Comparative anatomy of oil cells and mucilage cells in the leaves of the Lauraceae in China. Acta Phytotax Sin 37:529–540
Chu QG, Hu ZH (2001a) Developmental ultrastructure of oil and mucilage cells in Cinnamomum longepaniculatum. Sci Sin 37:19–25. https://doi.org/10.1111/j.1438-8677.1991.tb01564.x
Chu QG, Hu ZH (2001b) Ultrastructural studies on the development of oil cells in Litsea pungens. Acta Bot Sin 43:339–347
Coley PD, Barone JA (1996) Herbivory and plant defenses in tropical forests. Annu Rev Ecol Evol Syst 27:305–335. https://doi.org/10.1146/annurev.ecolsys.27.1.305
Costa ÉR, Louro GM, Simionatto S, Vasconcelos NG, Cardoso CA, Mallmann V, da Silva RCL, Matos MFC, Pizzuti L, Santiago EF, Morel AF, Mostardeiro MA, Simionatto E (2017) Chemical composition, antitumoral and antibacterial activities of essential oils from leaves and stem bark of Nectandra lanceolata (Lauraceae). J Essent Oil-Bear Plants 20(5):1184–1195. https://doi.org/10.1080/0972060X.2017.1394223
Coutinho LM (1978) O conceito de cerrado. Rev Bras Bot 1(1):17–23
Czaninskiy Y (1973) Observations sur une nouvelle couche pariétale dans les cellules associées aux vaisseaux du Robinier et du Sycomore. Protoplasma 77:211–219
David R, Carde JP (1964) Coloration différentielle dês inclusions lipidique et terpeniquesdes pseudophylles du pine maritime aumoyen du reactif Nadi, Paris. CR Acad Sci Paris D 257:1338–1340
De Barros TC, Leite VG, Pedersoli GD, Leme FM, Marinho CR, Teixeira SP (2023) Mucilage cells in the flower of Rosales species: reflections on morphological diversity, classification, and functions. Protoplasma 260:1135–1147. https://doi.org/10.1007/s00709-023-01836-8
Durigan G, Siqueira MFDE, Franco GADC (2007) Threats to the Cerrado remnants of the state of São Paulo, Brazil. Sci Agric 64:355–363
Ennajeh M, Vadel AM, Khemira H (2009) Osmoregulation and osmoprotection in the leaf cells of two olive cultivars subjected to severe water deficit. Acta Physiol Plant 31:711–721. https://doi.org/10.1007/s11738-009-0283-6
Espelie KE, Wattendorff J, Kolattukudy PE (1982) Composition and ultrastructure of the suberized cell wall of isolated crystal idioblasts from Agave americana L. leaves. Planta 155:166–175. https://doi.org/10.1007/BF00392548
Evert RF (2006) Esau’s plant anatomy: meristems, cells, and tissues of the plant body: their structure, function, and development. Wiley
Fahn A (1979) Secretory tissues in plants. Academic Press, London
Fahn A (1988) Secretory tissues in vascular plants. New Phytol 108:229–257. https://doi.org/10.1111/j.1469-8137.1988.tb04159.x
Fahn A, Cutler D (1992) Xerophytes. Gebruder Borntraeger, Berlin, Germany
Farago PV, Budel JM, Duarte MR, Nakashima T (2005) Análise morfoanatômica de folhas de Ocotea puberula (Rich.) Nees, Lauraceae. Rev Bras Farmacogn 15:250–255. https://doi.org/10.1590/S0102-695X2005000300016
Figueiredo ACS, Barroso JMG, Pedro LMG, Ascensão L (2007) Histoquímica e citoquímica em plantas: princípios e protocolos. 1.ed. Lisboa: Faculdade de Ciências da Universidade de Lisboa, Centro de Biotecnologia Vegetal, p 80
Foster AS (1956) Plant idioblasts: remarkable examples of cell specialization. Protoplasma 46:184–193
Freeman BC, Beattie AG (2008) An overview of plant defenses against pathogens and herbivores. Plant Health Instr 94. https://doi.org/10.1094/PHI-I-2008-0226-01
Geng SL, Cui ZX, Shu B, Zhao S, Yu XH (2012) Histochemistry and cell wall specialization of oil cells related to the essential oil accumulation in the bark of Cinnamomum cassia Presl. (Lauraceae). Plant Product Sci 15:1–9. https://doi.org/10.1626/pps.15.1
Gregory M, Baas P (1989) A survey of mucilage cells in vegetative organs of the dicotyledons. Isr J Plant Sci 38:125–174. https://doi.org/10.1080/0021213X.1989.10677119
Jensen WA (1962) Botanical histochemistry: principles and practice. W.H. Freeman and Co., San Francisco
Johansen DA (1940) Plant michrotechnique. McGraw-Hill, New York
Judd WS, Campbell CS, Kellogg EA, Stevens PF, Donoghue MJ (2008) Plants systematics: A phylogenetic approach. Sinauer Ass., Sunderland, MA
Kirk PW Jr (1970) Neutral red as a lipid fluorochrome. Stain Technol 45:1–4
Kraus JE, Arduin M (1997) Manual básico de métodos em morfologia vegetal. Editora Universidade Rural do Rio de Janeiro, Rio de Janeiro
Lusa MG, Cardoso EC, Machado SR, Appezzato-da-Glória B (2014) Trichomes related to an unusual method of water retention and protection of the stem apex in an arid zone perennial species. AoB Plants 7. https://doi.org/10.1093/aobpla/plu088
Machado SR, Canaveze Y, Rodrigues TM (2017) Structure and functioning of oil cavities in the shoot apex of Metrodorea nigra A. St.-Hil. (Rutaceae). Protoplasma 254:1661–1674. https://doi.org/10.1007/s00709-016-1056-x
Machado SR, Rodrigues TM (2021) Apoplasmic barrier in the extrafloral nectary of Citharexylum myrianthum (Verbenaceae). Planta 254:1–15. https://doi.org/10.1007/s00425-021-03663-8
Mariani P, Cappelletti EM, Campoccia D, Baldan B (1989) Oil cell ultrastructure and development in Liriodendron tulipifera L. Bot Gaz 150(4):391–396
Marinho CR, Zacaro AA, Ventrella MC (2011) Secretory cells in Piper umbellatum (Piperaceae) leaves: a new example for the development of idioblasts. Flora 206:1052–1062. https://doi.org/10.1016/j.flora.2011.07.011
Maron R, Fahn A (1979) Ultrastructure and development of oil cells in Laurus nobilis L. leaves. Bot J Linn Soc 78:31–40. https://doi.org/10.1111/j.1095-8339.1979.tb02184.x
Mastroberti AA, Mariath JE (2008) Development of mucilage cells of Araucaria angustifolia (Araucariaceae). Protoplasma 232:233–245. https://doi.org/10.1007/s00709-007-0274-7
Matthews ML, Endress PK (2006) Floral structure and systematics in four orders of rosids, including a broad survey of floral mucilage cells. Plant Syst Evol 260:199–221. https://doi.org/10.1007/s00606-006-0443-8
Mauseth JD (1980) A stereological morphometric study of the ultrastructure of mucilage cells in Opuntia polyacantha (Cactaceae). Bot Gaz 141:374–378
Mazia D, Brewer PA, Alfert M (1953) The cytochemical staining and measurement of protein with mercuric bromphenol blue. Biol Bull Rev 104:57–67
McNair JB (1918) Secretory canals of Rhus diversiloba. Bot Gaz 65(3):268–273. https://doi.org/10.1086/332233
Metcalfe CR, Chalk L (1950) Anatomy of the dicotyledons leaves, stem and wood in relation to taxonomy with notes on economy uses. Clarendon, Oxford
Metcalfe CR, Chalk L (1979) Anatomy of the Dicotyledons. v. 1: Systematic anatomy of leaf and stem, with a brief history of the subject. Clarendon: Oxford
O’Brien TP (1970) Further observations on hydrolysis of the cell wall in the xylem. Protoplasma 69:1–14
O’Brien TP, Feder N, McCully ME (1964) Polychromatic staining of plant cell walls by toluidine blue. Protoplasma 59:368–373
Oliveira PS, Marquis RJ (2002) The Cerrados of Brazil: Ecology and natural history of a Neotropical Savanna. Columbia University Press, New York
Paiva EAS, Oliveira DMT, Canaveze Y, Machado SR (2022) Adhesive secretion in Schizolobium parahyba (Vell.) Blake (Leguminosae: Caesalpinioideae): histochemical and morpho-functional characterization of this unusual feature in woody plants. Arthropod-Plant Interact 16:249–261. https://doi.org/10.1007/s11829-022-09888-y
Palermo FH, Rodrigues MIDA, de Nicolai J, Machado SR, Rodrigues TM (2018) Resin secretory canals in Protium heptaphyllum (Aubl.) Marchand. (Burseraceae): a tridimensional branched and anastomosed system. Protoplasma 255:899–910. https://doi.org/10.1007/s00709-017-1197-6
Pate JS, Gunning BES (1972) Transfer cells. Ann Rev Plant Physiol 23:173–196
Pizzolato P, Lillie RD (1973) Mayer’s tannic acid-ferric chloride stain for mucins. J Histochem Cytochem 21:56–64
Płachno BJ, Kapusta M, Światek P, Stolarczyk P, Kocki J (2020) Immunodetection of petic epitopes, arabinogalactan proteins, and extensins, in mucilage cells from the ovules of Pilosella officinarum Vaill. and Taraxacum officinale Agg. (Asteraceae). Int J Mol Sci 21(24):9642. https://doi.org/10.3390/ijms21249642
Platt-Aloia KA, Oross JW, Thomson WW (1983) Ultrastructural study of the development of oil cells in the mesocarp of avocado fruit. Bot Gaz 144:49–55
Ramakrishna A, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6:1720–1731. https://doi.org/10.4161/psb.6.11.17613
Ratter JA, Ribeiro JF, Bridgewater S (1997) The Brazilian cerrado vegetation and threats to its biodiversity. Ann Bot 80:223–230. https://doi.org/10.1006/anbo.1997.0469
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Rocha JF, Pimentel RR, Machado SR (2011) Estruturas secretoras de mucilagem em Hibiscus pernambucensis Arruda (Malvaceae): distribuição, caracterização morfoanatômica e histoquímica. Acta Bot Bras 25:751–763. https://doi.org/10.1590/S0102-33062011000400003
Rodriguez-Saona C, Trumble JT (2003) Secretory avocado idioblast oil cells: evidence of their defensive role against a non-adapted insect herbivore. Entomol Exp Appl 94:183–194
Rohwer J (1993) Lauraceae. In: Kubitzki K, Rohwer JG, Bittrich V, editors. The families and genera of vascular plants. II; Dicotyledons: Magnoliid, Hamamelid and Caryophyllid Families. Springer Verlag, Berlin, pp 366–91
Roshchina VV, Roshchina VD (1993) The excretory function of higher plants. Springer, Berlin
Sajo MDG, Machado SR (2001) Submicroscopical features of leaves of Xyris species. Braz Arch Biol Technol 44:405–410. https://doi.org/10.1590/S1516-89132001000400011
Scott FM, Bystrom BG, Bowler E (1963) Persea americana, mesocarp cell structure, light and electron microscope study. Bot Gaz 124:423–428
Solereder H (1908) Systematic anatomy of the Dicotyledons: A handbook for laboratories of pure and applied botany, vol 2. Clarendon Press, Oxford
Teixeira SP, Machado SR (2007) Glandular dots of Caesalpinia echinata Lam. (Leguminosae): distribution, structure and ultrastructure. J Torrey Bot Soc 134:135–143. https://doi.org/10.3159/1095-5674
Teles MMRS, Pinheiro AAV, Dias CDS, Tavares JF, Barbosa Filho JM, Da Cunha EVL (2019) Alkaloids of the Lauraceae. Alkaloids: Chem Biol 82:147–304
Tolera M, Menger D, Sass-Klaassen U, Sterck FJ, Copini P, Bongers F (2013) Resin secretory structures of Boswellia papyrifera and applications for franckincense yield. Ann Bot 111(1):61–68. https://doi.org/10.1093/aob/mcs236
Tresmondi F, Nogueira A, Guimarães E, Machado SR (2015) Morphology, secretion composition, and ecological aspects of stipular colleters in Rubiaceae species from tropical forest and savanna. Sci Nat 102:73. https://doi.org/10.1007/s00114-015-1320-5
Wisniewski M, Davis G (1989) Evidence for the involvement of a specific cell wall layer in regulation of deep supercooling of xylem parenchyma. Plant Physiol 91:151–156. https://doi.org/10.1104/pp.91.1.151
Xia X, Zhang HM, Offler CE, Patrick JW (2017) A structurally specialized uniform wall layer is essential for constructing wall ingrowth papillae in transfer cells. Front Plant Sci 8:2035. https://doi.org/10.3389/fpls.2017.02035
Zhang Y, Cai P, Cheng G, Zhang Y (2022) A brief review of phenolic compounds identified from plants: Their extraction, analysis, and biological activity. Nat Prod Commun 17:1934578X211069721. https://doi.org/10.1177/1934578X211069721
Acknowledgements
We are grateful to specialist Dr João Batista Baitello from Instituto Florestal de São Paulo, Brazil, for plant species identification; to Dr Shelly Favorito Carvalho for help with fluorescence methods; to the Electron Microscopy Center at the Institute of Bioscience (UNESP) for assistance in processing the materials and the provision of equipment for electron microscopy; and to Dr. Sandy Lang (Rescript.co.nz) for helping with the review and editing of the English language.
Funding
This work was supported by the ‘Coordination for the Improvement of Higher Education Personnel’—Brazil (CAPES), Finance Code 001, and by the São Paulo Research Foundation (FAPESP Grant #2021/13392–0) to SRM. SRM [Grant number 308982/2020–7] receives scholarships from the National Council for Scientific and Technological Development (CNPq).
Author information
Authors and Affiliations
Contributions
Silvia Rodrigues Machado and Karla Bianca de Deus Bento conceived and designed the research. Karla Bianca de Deus Bento and Yve Canaveze carried out the laboratory analyses. All authors carried out the data analyses and wrote the manuscript.
Corresponding author
Ethics declarations
The authors have no relevant financial or non-financial interests to disclose that are directly or indirectly related to the work submitted for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Alexander Schulz
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Deus Bento, K.B., Canaveze, Y. & Machado, S.R. Oil and mucilage idioblasts co-occur in the vegetative organs of Ocotea pulchella (Lauraceae): comparative development, ultrastructure and secretions. Protoplasma (2024). https://doi.org/10.1007/s00709-024-01942-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00709-024-01942-1