Abstract
Plant-pollinator interactions are generally mutualistic, but if one of the partners can get rewards without providing any services in return, these interactions become antagonistic. In the oil flower syndrome, the highly specialized system suggests a high degree mutualism; however, there are reports of illegitimate oil-collecting bees in flowers of Malpighiaceae. We evaluated the legitimate and illegitimate visits to flowers in 15 species of this plant family along a latitudinal gradient in Argentina. Our results show that oil robbery in most of the Malpighiaceae analyzed species was as common as the legitimate collection of this resource by oil-collecting bees, and significant differences in the proportion of illegitimate visits along the latitudinal gradient, being the robbery more common to southern sites.
Similar content being viewed by others
1 Introduction
Flowers attract and reward their pollinators so that they transport their pollen grains to another compatible co-specific flower and enable the pollination and posterior sexual reproduction of the species. To achieve pollen effective transference, pollinators must legitimately forage, contacting fertile pieces in successive floral visits. This pollinator behavior leads to mutualistic interactions (with benefits for both partners) and is known as loyalty. Also, the term “legitimate” has been used for flower visitors that appear to be adapted to host flowers (Inouye 1981). However, in the plant-pollinator interactions, one of the partners can get rewards without providing any services in return; therefore, these mutualistic interactions become antagonistic. In pollination ecology, the pollinator attraction without offering a reward leads to pollination by deception (Proctor et al. 1996), and plants exhibit distinct ways of dishonesty, as sexual, oviposition substrate, and food deception, among others (Dafni 1984; Johnson and Schiestl 2016). On the other hand, this antagonistic behavior in animal partners is known as robbery, and in these cases, flower visitors are called “illegitimate” (Inouye 1981). Numerous animal species are nectar and/or pollen robbers (Faegri and van der Pijl 1966, Inouye 1981, but see Maloof and Inouye 2000). However, little is known about the robbers of other (more specialized) rewards, as floral oil (Vogel 1990; Sigrist and Sazima 2004; Barônio et al. 2020; Maubecin et al. 2021) or floral perfumes (Boff et al. 2015; Reposi et al. 2021).
Floral oils are produced by diverse plant species (Renner and Schaefer 2010) in glandular structures called elaiophores (Vogel 1974), which exist in two types: epithelial and trichomatic (Vogel 1974), with some species showing intermediate elaiophores (Gomiz et al. 2013). Oil-rewarding plants occur both in the New and the Old World, but are most diverse in the Neotropical region (Renner and Schaefer 2010), where they are represented by species in seven plant families: Malpighicaeae, Iridaceae, Plantaginaceae, Solanaceae, Krameriaceae, Calceolariaceae, and Orchidaceae (ordered in a probable chronological sequence of occurrence of oil flower syndrome; Renner and Schaefer 2010). Also, some species of Melastomataceae are recognized as oil-rewarding species (Buchmann 1987; Oliveira et al. 2022.) As counterpart, in the Neotropical region, oil-collecting bees are represented by three tribes in the family Apidae: Centridini, Tapinotaspidini, and Tetrapediini (Michener 2007). These bees are equipped with specialized setal combs and pads, which structure and position vary among different oil-collecting groups (Neff and Simpson 1981; Roig-Alsina 1997; Cocucci et al. 2000). Floral oils are used by female bees for nest construction and protection and/or mixed with pollen mass for larval food (Vinson et al. 1996). Also, males of Paratetrapedia Moure and Tetrapedia Klug have a morphological structure for oil collection and some species used this floral oil (Cappellari et al. 2012). However, the role of floral oils in males is still unknown (Cappellari et al. 2012; Danforth et al. 2019).
The family Malpighiaceae is arguably the oldest lineage to have acquired oil glands (Renner and Schaefer 2010), and most of the Neotropical species (~ 1000) produce floral oils in epithelial elaiophores, which appear paired in the abaxial surface of four or five sepals on the calyx (Vogel 1974; Anderson 1979). These glands have a common morpho-anatomical pattern, even though they show some differences in number by flower, morphology, and/or anatomy among the species (Aliscioni et al. 2021). Neotropical Malpighiaceae species present a floral conservatism related to attraction, orientation, and reward (Anderson 1979), but they vary in type of inflorescences and number of synchronically opened flowers. Females of most species of Centris Fabricius and all species of Epicharis Klug (Centridini) and Monoeca Lepeletier and Serville (Tapinotaspidini) have oil-collecting organs on four legs adapted for oil harvesting on these flowers. Moreover, combs in these species are arranged in an anteroventral (or inner) position in the fore- and middle legs, in a “four-legged” pattern of the oil-collecting organs (Neff and Simpson 1981). To gather oil with its fore- and mid-legs from the elaiophores, the females grasp the base of the flag petal with its mandibles and scrap the elaiophores (Figure 1a–d). During this stereotyped oil-collecting behavior, these bees contact the ventral area of the body with anthers and/or stigmas (Figure 1a–d). This pollinator behavior is necessary in flowers of Malpighiaceae, principally due to the requirement of mechanical rupture of a thick stigmatic cuticle that is produced by the oil-collecting bees (Sigrist and Sazima 2004; Aliscioni et al. 2018, 2019). Moreover, the rupture of connective glands in heterantherous (i.e., presence of stamens with more than one type of anther in the same flower) species could be a necessary mechanism for maximizing the transport of pollen grains (Avalos et al. 2020). The rupture of the stigmatic cuticle exposes the stigmatic surface, and promotes the secretion of stigmatic exudates that are accumulated under cuticle, which allow adherence, hydration, and germination of pollen grains (Sigrist and Sazima 2004; Aliscioni et al. 2018). On the other hand, Avalos et al. (2020) showed in two heterantherous species of Stigmaphyllon that the connective glands of a set of anthers produce large quantities of mucilage, which would improve pollen transport (increasing adherence to pollinator body and dampness maintenance), and these authors suggested a necessary rupture of these glands for mechanical action of pollinators (Avalos et al. 2020).
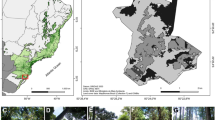
Loyalty in floral visits to Malpighiaceae species. a b Centris proxima performing the stereotyped visit on a Stigmaphyllon bonariense flower. a Before landing on the flower and b during oil collection, note that its mandibles grasp the flag petal (fp) claw (black arrow), fore- and middle legs scrape the elaiophores (white arrows), and the ventral part of the mesosoma contacts with reproductive structures (asterisk). c Epicharis analis legitimately visiting a flower of S. bonariense. d Monoeca armata legitimately visiting a flower of Heteropterys intermedia, note that its fore- and middle legs scrape the elaiophores (white arrows) and its head is correctly oriented towards the flag petal (fp).
All species of Epicharis use floral oil of Malpighiaceae, but the species of Centris exhibit high variability of host plants (Martins et al. 2015), and some lineages have lost the four-legged oil-collecting apparatus (Neff and Simpson 1981; Martins et al. 2015). The tribe Tapinotaspidini is the most diverse in number of genera and variety of oil-collecting adaptations (Roig-Alsina 1997; Cocucci et al. 2000). These bees have peculiar groups of specialized hairs present on the forelegs, middle legs, or metasomal sterna (Roig-Alsina 1997). Most genera of Tapinotaspidini are oil collectors in flowers with trichome elaiophores (Roig-Alsina 1997; Cocucci et al. 2000), but Monoeca exploits epithelial elaiophores and some species of Arhysoceble Moure, and Paratetrapedia s.l. use different types of oil glands (Vogel and Cocucci 1995; Cocucci et al. 2000; Gomiz et al. 2014; Neff and Simpson 2017; Torretta and Roig-Alsina 2017; Torretta et al. 2017; Aguiar et al. 2020). In species of Tapinotaspidini with oil-collecting specialized hairs on forelegs, the collecting apparatus is located posteroventrally (or externally), in a “two-legged” pattern of the oil-collecting apparatuses (Neff and Simpson 1981). Finally, females and males of Tetrapedia spp. have combs on the forebasitarsi located posteroventrally (or externally), in a “two-legged” pattern of the oil-collecting organs. However, the tarsal combs are turned distally in Tetrapedia, and slightly differ in their anterior position from those of Tapinotaspidini (e.g., Paratetrapedia s.l., Neff and Simpson 1981). Therefore, bees of these species of Tapinotaspidini and Tetrapediini, to forage for floral oil, perch on the sepals outside the flower and scrape the elaiophores (Figure 2a–c), making it impossible to contact the fertile pieces (anther and/or stigmas). These species exhibit different behavior for oil collection than that used by Centris, Epicharis, and Monoeca species (Vogel 1974).
Robbery of floral oil and pollen-collecting in Malpighiaceae species. a–c Robbery in anthetic flowers. a Paratetrapedia (L.) nigrispinis on a flower of Stigmaphyllon jatrophifolium, b Tetrapedia sp. on a flower of S. bonariense, and c Caenonomada bruneri on a flower of Heteropterys glabra; all species collecting oils with forelegs (black arrows) without contacting the fertile floral pieces (white arrows). d Pollen collection. d Paratetrapedia (L.) nigrispinis collecting pollen from a flower of Janusia guaranitica with forelegs (white arrow) and transporting on the scopa (black arrow), the flag petal (fp) is indicated. e–f Robbery in pre-anthetic flowers. e Centris proxima collecting oil with fore- and middle legs (black arrows) from floral bud of S. bonariense. f Tetrapedia sp. collecting oil with forelegs (black arrow) from the floral bud of Banisteriopsis muricata, note that its mandibles grasp the floral receptacle.
In the Neotropical region, the family Malpighiaceae reaches the highest diversity in the tropical areas, and in Argentina, the maximum richness occurs in the northern province of Misiones, decreasing markedly southwards (Aliscioni and Torretta 2017). Similar patterns of species richness are observed in oil-collecting bees associated with Malpighiaceae: Centris (Roig-Alsina 2000), Epicharis (Moure et al. 2007), Monoeca (Torretta and Roig-Alsina 2016), and Paratetrapedia s.l. (Torretta and Roig-Alsina 2017). Hence, the richness of Malpighiaceae which these bee species can collect floral oil among sites could be different. In the northern sites, species of oil-collecting bees can visit a higher number of Malpighiaceae species, while in the southernmost sites, the number of species of this plant family is reduced; thus, the oil-collecting bees depend on a lesser number of Malpighiaceae species at the populations closer to the southern border (Torretta et al. 2017).
Interactions between oil flowers and oil-collecting bees are highly specialized, and suggest a high degree of mutualism. Therefore, we hypothesized that loyalty (i.e., legitimate visits) should be the rule among these bees and their host plants. However, due to reports of illegitimate oil-collecting bees in flowers of Malpighiaceae (Vogel 1974, Sigrist and Sazima 2004, Rocha-Filho and Garófalo 2016, Torretta et al. 2017a, Aguiar et al. 2020, among others), our principal objective is to quantify how common are oil robbers in the flowers of Malpighiaceae in Argentina. To achieve this, we evaluated this hypothesis in 15 species of Malpighiaceae along a latitudinal gradient in Argentina. We predicted that (a) the visitation rate of legitimate pollinators (loyalty) is greater than the visitation rate of illegitimate robbers (robbery) and (b) the relation of legitimate vs. illegitimate visits varied with latitude.
2 Materials and methods
2.1 Study sites
Natural populations of 15 species of Malpighiaceae (Table I) were selected in 22 different sites in northeastern to central Argentina (Figure 3) along a latitudinal gradient (across more than 1000 km) that includes different units of vegetation for Argentina (Oyarzabal et al. 2018). These areas include tropical rain forests, riverine forests, lowland forests, floodplains, public parks, and other modified environments, with different richness of co-flowering species of Malpighiaceae. Sampling was carried out during five consecutive years, but not all sites were visited on each occasion (Dec/Mar 2012/2013, Dec/Feb 2013/2014, Nov/Feb 2014/2015, Nov/Dec 2015, and Dec/Apr 2016/2017, Table I). Each fieldwork period consisted of 1–3 days of observations, censuses, and capture of oil-collecting bees while foraging for this floral resource. Because our objective was to evaluate the loyalty and robbery during visits for floral oil collection, we have not taken into account visits for pollen collection.
2.2 Assemblages of legitimate and illegitimate oil-collecting floral visitors
We sampled species of oil-collecting bees across populations on different days (totalizing 1–6 days per population) and at different times of the day (between 800 and 1900 h). We conducted 284 censuses of a duration of 10 min on a known number of flowers (cumulative time = 47.3 h, Table I). With these data, we calculated the floral visitors for each species of Malpighiaceae, discriminating between legitimate (i.e., loyalty) and illegitimate (i.e., robbery) visitors during the stereotyped behavior of oil collection. Species that were observed making contact with reproductive structures while foraging for floral oil (i.e., four-legged pattern) were recorded as legitimate pollinators, in such a way, we discriminate pollinators from oil-robber visitors. We included in this last category to robber and thief visitors (Inouye 1981; Barônio et al. 2020).
2.3 Statistical analysis
In order to know whether the legitimate visits are the rule in this highly specialized system, we performed a generalized linear mixed model (GLMM). We used as a response variable the difference in the number of visits of pollinators and oil-robber bees (legitimate vs. illegitimate visitors) in each census. Each value is the difference in the frequency of visits made by legitimate and illegitimate visitors during a 10-min census of a plant species on a known number of flowers. The GLMM was performed using the lmer function (lme4 package, R Development Core Team 2013) with Gaussian distributions. The population and the date of the survey were used as random factors, while the plant species were used as a fixed factor (see model specifications). In this model (M1), the intercept was removed in order to estimate if the mean of the differences were different from zero (Table S1). Positive values indicate a greater visitation rate of legitimate visitors than illegitimate ones, while negative values indicate the opposite.
On the other hand, in order to estimate if there are differences in the visitation between legitimate and illegitimate visitors related to the latitude where the surveys were carried out, we performed another GLMM using the glmer function with binomial distributions. In this model (M2), the response variable was a matrix with two columns, with the first column being the number of legitimate visitors and the second being the number of illegitimate visits during each 10-min census on a known number of flowers of each Malpighiaceae species (Table S1). We considered the pollinator visit as “success” and the oil-robber visit as “failure.” In the binomial model, we used the cbind function to indicate the response variable instead of using the proportion values (Mangiafico 2015). The plant species and the date of the survey were used as random factors, while the latitude of the sites and the number of flowers (log-transformed) observed in each census were used as fixed factors (see model specifications). For both models, only censuses with visits were considered. The map indicating the studied populations was constructed using SimpleMappr (Shorthouse 2010).
3 Results
3.1 Floral visitor richness and number of visits
In total, we registered 323 oil-collecting bees on flowers of 15 species of Malpighiaceae (Table II). Of them, 183 individuals (17 species) were legitimate visitors: Centris (11 spp.), Epicharis (4 spp.), and Monoeca (2 spp.) (Table II); and 140 individuals (13 species) were illegitimate visitors: Arhysoceble (1 sp.), Caenonomada Ashmead (1 sp.), Paratetrapedia (5 spp.), and Tetrapedia (6 spp.) (Table II). The number of visits per census showed a negative exponential distribution (Figure 4), with 42.2% of the census without visits, 52.5% with few (1–3) visits, and the remainder 5.3% with more than 3 visits (up to 8) in 10 min of observation.
3.2 Loyalty and robbery in floral visits
The frequency of visits made by legitimate and illegitimate visitors during the censuses did not show significant differences in 12 of the 15 analyzed species, while Stigmaphyllon bonariense and S. jatrophifolium were significantly more visited by legitimate pollinators and Janusia guaranitica exhibited the opposite pattern (Figure 5, Table S2).
Histogram (with density line) of the relation between legitimate versus illegitimate visits in flowers of the Malpighiaceae studied species. The density of the data in the histogram is a smoothed-out representation of the distribution. The vertical dashed blue line indicates the mean value. Density lines: blue indicates more visits of pollinators than illegitimate floral visitors; yellow indicates the opposite pattern; and black indicates not significant differences. Note *p < 0.05; **p < 0.005.
3.3 Differences in the visitation rates of pollinators and oil-robbery bees related to the latitude
We found significant differences among the proportion of pollinators and oil-robbery bees in relation to latitude (z = − 2.933, p = 0.00336, Table S3, Figure S1a), being higher the proportion of the illegitimate interactions in the southern sites. These differences are dependent on the latitude (z = 2.145, p = 0.03193, Table S3) and the log-transformed number of flower (z = 2.411, p = 0.0159, Table S3, Figure S1b).
4 Discussion
Due to the high specialization between oil-rewarding flowers (e.g., elaiophore type) and oil-collecting bees (e.g., morphology and position of collector apparatus), we hypothesized that loyalty should be the rule in these interactions. However, our results are contrary to our expectations (prediction a), and show that oil robbery in most of the Malpighiaceae analyzed species was as common as the legitimate collection of this resource by oil-collecting bees. The gathering of floral oils for species of Tapinotaspidini and Tetrapediini has been reported for many species of Malpighiaceae (Vogel 1974; Sigrist and Sazima 2004; Aguiar 2009; Aguiar and Melo 2011, Rocha-Filho and Garófalo 2016; Torretta al. 2017; Aguiar et al. 2020; Aliscioni et al. 2021, among others). Even though these interactions were reported as antagonistic (Neff and Simpson 1981; Steiner 1985; Vogel 1990; Sigrist and Sazima 2004), only one study focused on antagonistic interactions (Barônio et al. 2020). Our work statistically analyzed for the first time the loyalty and robbery in the interactions among the oil flowers of Malpighiaceae and its oil-collecting bees. As we mentioned, due to the posteroventral position of the tarsal combs in these bee species, in order to collect the floral oil, the individuals crawled underneath the flower, where they scraped the elaiophores (Figure 2a–c). Vogel (1990) suggested that some species of Tapinotaspidini and Tetrapediini might be legitimate pollinators of small-flowered genera of Malpighiaceae, while pollen-collecting (Figure 2d). However, if the rupture of the stigmatic cuticle is a fundamental requisite for an efficient pollination in most Malpighiaceae species (Sigrist and Sazima 2004; Aliscioni et al. 2018, 2019), and if these oil-collecting bees (in general, smaller and more delicate than species of Centris and Epicharis) fail to break the cuticle, their quality as pollinator should be analyzed in each focal species. Steiner (1985) suggested that Spachea membranacea Cuatrec. and Malpighia romeroana Cuatrec., two small-flowered species of Central America understory, are pollinated by species of Paratetrapedia, which are legitimate pollen-collecting and oil-robber bees. Therefore, independently of their efficiency as a pollinator of species of Malpighiaceae, oil-collecting bees of genera Paratetrapedia s.l., Arhysoceble, Caenonomada (Tapinotaspidini), and Tetrapedia (Tetrapediini) are oil robbers on flowers of this plant family. It is noticeable that these tapinotaspidine genera exclusively visit flowers of species of Malpighiaceae, or visit flowers of species of Malpighiaceae and from species of other families (e.g., Iridaceae, Krameriaceae, Orchidaceae, Plantaginaceae) (Aguiar and Melo 2009; Gomiz et al. 2014; Torretta and Roig-Alsina 2017; Carneiro et al. 2019; Aguiar et al. 2020), acting as a legitimate pollinator in non-Malpighiaceae families (Gomiz et al. 2014; Martins et al. 2016). These other families present oil glands in other floral pieces (not in the abaxial surface of the sepals), and the bees contact the anthers and/or stigmas during the legitimate oil-collecting behavior. The position of Monoeca as sister to all remaining Tapinotaspidini (Roig-Alsina 1997; Aguiar et al. 2020) suggests the use of epithelial elaiophores as a primitive condition in Tapinotaspidini, with posterior changes to trichomatic elaiophores in the other genera. As we have mentioned, such changes can be partial or total (e.g., species of Chalepogenus Holmberg, Lanthanella Michener and Moure, and Lanthanomelissa Holmberg exclusively oil-foraging from trichomatic elaiophores) (Neff and Simpson 1981; Roig-Alsina 1997; Cocucci et al. 2000). On the other hand, the oil-collecting structures of Tetrapedia appear to be adapted for harvesting oils from epithelial elaiophores (Neff and Simpson 1981) as found in the Malpighiaceae, and some Oncidiinae orchids (Singer and Cocucci 1999); however, there are also reports of females and males collecting oil from trichomatic elaiophores from Plantaginaceae (Vogel and Machado 1991), Iridaceae (Alves-dos-Santos 1999) and trichomatic and intermediate as in other Oncidiinae orchids (Reis et al. 2006; Gomiz et al. 2017). Possibly, the Tetrapedia species are the most versatile among the all oil-collecting bees, with respect to the type of elaiophore they use.
Barônio et al. (2020) studied the oil robbery by small oil-collecting bees in Malpighiaceae flowers, focusing on the role of upside-down behavior performed by two Tetrapedia species (defined by them as “small oil-collecting bees”) in Peixotoa tomentosa, a species with large yellow flowers. These authors mentioned that “there is a wide variation in body size among oil-collecting bee species (Centridini, Tapinotaspidini, and Tetrapediini), which determines the coupling capacity of bees to flowers” but they did not mention anything about the position (inner or outer) of oil-collecting organs on two or four legs. For example, some species of Tetrapedia (tarsal combs outer in a two-legged pattern of the oil-collecting organs) and Monoeca (tarsal combs inner in a four-legged pattern of the oil-collecting organs) are similar in sizes; however, they differ in their oil-collecting behaviors. The position of the oil-collecting organs, and not the body size of bees, determines the loyalty in the oil gathering in the oil-collecting bees. In Argentina, several species of Centris and Epicharis, with different sizes (from medium-sized [⁓1.5 cm long] to very large-sized bees [3–3.5 cm long]) visit Malpighiaceae flowers of different sizes (Torretta et al. 2017; Aliscioni et al. 2021; Avalos et al. 2021) with different pollination efficiency (i.e., contacting one, two or all three stigmas per visit; Aliscioni et al. 2018; Avalos et al. 2021); however, all species legitimately collect the floral oil. Furthermore, the Argentine species of Monoeca legitimately forage for oil and use the pollen of small-flowered Malpighiaceae (Torretta and Roig-Alsina 2016).
Only two of our studied species (Stigmaphyllon bonariense (Hook. and Arn.) C.E. Anderson and S. jatrophifolium A. Juss.) received more visits from pollinators than illegitimate floral visitors. Both species are common woody lianas, with large yellow flowers (2.5–3 cm), wide distributions within Argentina (Torretta et al. 2017), and prolonged flowering phenologies (November to April/May, Avalos et al. 2021). In these species, there is a marked turnover in the pollinator assemblage throughout the flowering season (Avalos et al. 2021) with the predominance of both Centris and Epicharis (Torretta et al. 2017). Flowers of these species are among the largest of the Argentine Malpighiaceae and are visited by medium (e.g., Centris trigonoides Lepeletier), large (e.g., Epicharis affinis Smith), and very large (e.g., Centris obsoleta Lepeletier) oil-collecting bees. A possible explanation for the higher proportion of legitimate visits compared to the illegitimate ones is that its large flowers offer high quantities of reward that satisfy larger pollinators (Cohen and Shmida 1993), and these bees displace to the smaller ones of tribes Tapinotaspidini and Tetrapediini, which are more abundant at the beginning of the flowering season (Avalos et al. 2021). Another possible explanation (non-mutually exclusive) is that Paratetrapedia s.l., and Tetrapedia species preferentially visit a wide variety of Malpighiaceae with small flowers, reducing their visits to large-flowered species in presence of Centridini bees. The two Argentine Monoeca species, which could legitimately collect floral oil in these species, were not observed in Stigmaphyllon flowers, possibly due to their great size.
Conversely, Janusia guaranitica was the only species more visited by illegitimate than legitimate floral visitors in our censuses. This species is a scandent shrub, with wide distribution in Argentina, that mainly occurs in open habitats (Aliscioni and Torretta 2017) where the diversity of Centris and Epicharis is diminished with respect to forested areas in Argentina (Roig-Alsina 2000). We observed only three Centris species legitimately oil-collecting in their flowers (C. tarsata Smith, C. trigonoides, and C. tricolor Friese) and six oil-robber bee species (Arhysoceble picta (Friese), Caenonomada bruneri Ashmead, Paratetrapedia (Lophopedia) nigrispinis (Vachal), P. (Paratetrapedia) leucostoma (Cockerell) and two unidentified species of Tetrapedia). Of the latter, some species of Paratetrapia and Tetrapedia (Avalos and Torretta, obs. pers.) were seen legitimately collecting pollen (Figure 2d). Therefore, the low diversity of Centris species and the dual resource collection (oil and pollen) could be explaining the higher rate of visits of these illegitimate oil-collecting bees.
In relation to the variation in the proportion of illegitimate visits along the latitudinal gradient (prediction b), our results exhibit significant differences being the robbery more common in southern sites than in northernmost ones. In our southernmost sites, we registered only three oil-robbery bee species (Caenonomada bruneri, Paratetrapedia nigrispinis, and one undetermined species of Tetrapedia), being the last two abundant (Torretta obs. pers.). In these sites, the richness of Malpighiaceae is low (two or three species), and although there are other oil-rewarding species belonging to families Iridaceae (Cypella, Sisyrinchium) and Solanaceae (Nierembergia), these latter species are not visited for Paratetrapedia nigrispinis and Tetrapedia sp. (Cocucci 1991; Roig-Alsina 1999; Cocucci and Vogel 2001; Torretta et al. 2011). This fact generates a great dependency of these oil-robbery bee species for the Malpighiaceae species to obtain this resource. Due to these variations along the latitudinal gradient, co-specific individuals occurring in different populations might suffer differential positive/negative selection promoted by their interactions with legitimate and illegitimate floral visitors.
The oil robbery by legitimate pollinators deserves a special mention. It is known that several species of oil-collecting bees harvest oil in floral buds (Figure 2e; Vogel 1974; Vinson et al. 1997; Vilas Boas et al. 2013; Neff and Simpson 2017; de Melo et al. 2018). However, this behavior is not the rule. If so, the elaiophores of the anthetic flowers would have a broken cuticle and lesser availability of oil, which would make them less attractive to pollinators. The collection of oil in floral buds could indicate that this reward is a limiting resource for these species, and when the availability of flowers at anthesis is low and/or the abundance of females is high, these legitimate oil-collecting bees behave like robbers, gathering floral oil from the buds. Also, species with a “two-legged” pattern of the oil-collecting apparatuses obtained this resource from buds (Figure 2f). Among all Neotropical oil-rewarding plant families, flowers of Malpighiaceae are the only ones in which the elaiophores are exposed at the bud stage, due to its position in the abaxial epidermis at the sepals (Figure 2e–f); in species of the other families (Calceolariaceae, Iridaceae, Krameriaceae, Orchidaceae, Plantaginaceae, and Solanaceae), the elaiophores are covered by the unfolded corolla in pre-anthetic flowers. Therefore, the oil robbery in floral buds could be exclusive to species of Malpighiaceae.
5 Conclusion
We conclude that in Malpighiaceae species in our study area the occurrence of legitimate interactions (i.e., loyalty) is similar to illegitimate ones (robbery). Still, even though these highly specialized interactions suggest a high degree of mutualism, loyalty seems not to be the rule. However, very little is known about oil production dynamics in flowers of Malpighiaceae (i.e., continued production throughout the floral life, total production in pre-anthesis, etc.) (Vogel 1974; de Melo et al. 2018); therefore, the net effects of these illegitimate interactions need to be studied. We hope that our results will trigger new questions to reliably understand these interesting mutualistic/antagonistic interactions.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Aguiar AJC (2009) Taxonomic revision of the bee genus Lophopedia Michener and Moure (Hymenoptera, Apidae, Tapinotaspidini). Zootaxa 2193:1–52
Aguiar AJC, Melo GAR (2011) Revision and phylogeny of the bee genus Paratetrapedia Moure, with description of a new genus from the Andean Cordillera (Hymenoptera, Apidae, Tapinotaspidini). Zool J Linn Soc-Lond 162:351–442
Aguiar AJC, Melo GAR, Vasconcelos TNC, Gonçalves RB, Giugliano L, Martins AC (2020) Biogeography and early diversification of Tapinotaspidini oil-bees support presence of Paleocene savannas in South America. Mol Phylogenet Evol 143:106692
Aliscioni SS, Torretta JP (2017) Malpighiaceae, in: Zuloaga, F. O. and Belgrano, M. J. (Eds.), Flora Vascular de la República Argentina, vol. 17. Estudio Sigma S.R.L., Buenos Aires, pp. 163–205
Aliscioni SS, Gotelli M, Torretta JP (2018) Structure of the stigma and style of Callaeum psilophyllum (Malpighiaceae) and its relation with potential pollinators. Protoplasma 255:1433–1442
Aliscioni SS, Gotelli M, Torretta JP (2019) Carpel dimorphism in gynoecium of Tricomaria usillo, and comparison with other genera of the Carolus clade (Malpighiaceae). Protoplasma 256:1133–1144
Aliscioni SS, Gomiz NE, Agüero JI, Torretta JP (2021) Structural diversity of elaiophores in Argentine species of Malpighiaceae: morphology, anatomy and interaction with pollinators. Protoplasma 259:789–807
Alves-dos-Santos I (1999) Abelhas e plantas melíferas da Mata Atlântica, restinga e dunas do litoral norte do estado do Rio Grande do Sul. Brasil Revta Bras Entomol 43(3/4):191–223
Anderson WR (1979) Floral conservatism in Neotropical Malpighiaceae. Biotropica 11:219–223
Avalos AA, Torretta JP, Lattar EC, Ferrucci MS (2020) Structure and development of the anthers and connective glands in two species of Stigmaphyllon (Malpighiaceae): are the heteromorphic anthers related to division of labour? Protoplasma 257:1165–1181
Avalos AA, Marrero HJ, Ferrucci MS, Torretta JP (2021) Stigmas arrangement, reproductive system and maternal reproductive success in two species of Stigmaphyllon (Malpighiaceae): does the size of pollinators matter? Plant Ecol 222(11):1263–1279
Barônio GJ, de Brito VLG, Nogueira A, Rech AR (2020) Beyond robbery: the role of upside-down behaviour performed by small oil-collecting bees in Malpighiaceae. Arthropod-Plant Interact 14:613–621
Boff S, Demarco D, Marchi P, Alves-dos-Santos I (2015) Perfume production in flowers of Angelonia salicariifolia attracts males of Euglossa annectans which do not promote pollination. Apidologie 46:84–91
Buchmann SL (1987) The ecology of oil flowers and their bees. Annu Rev Ecol Syst 18:343–369
Cappellari SC, Melo GAR, Aguiar AJC, Neff JL (2012) Floral oil collection by male Tetrapedia bees (Hymenoptera: Apidae: Tetrapediini). Apidologie 43:39–50
Carneiro LT, André CBDS, Takahasi A, Alves-dos-Santos I (2019) Interactions between oil-collecting bees and Krameria grandiflora (Krameriaceae) with emphasis on the role of specialized floral traits in the mutual fit. Arthropod-Plant Interact 13(2):213–226
Cocucci AA (1991) Pollination biology of Nierembergia (Solanaceae). Plant Syst Evol 174(1):17–35
Cocucci AA, Vogel S (2001) Oil-producing flowers of Sisyrinchium species (Iridaceae) and their pollinators in southern South America. Flora 196(1):26–46
Cocucci AA, Sérsic A, Roig-Alsina AH (2000) Oil-collecting structures in Tapinotaspidini: their diversity, function and probable origin (Hymenoptera: Apidae). Mitt Münch Entomol Ges 90:51–74
Cohen D, Shmida A (1993) The evolution of flower display and reward. Evol Biol 27:197–243
Dafni A (1984) Mimicry and deception in pollination. Annu Rev Ecol Evol Syst 15:259–278
Danforth BN, Minckley RL, Neff JL (2019) The solitary bees: biology, evolution, conservation. Princeton University Press, Princeton
de Melo BT, Mota T, Schlindwein C, Antonini Y, Oliveira R (2018) Floral colour change in Byrsonima variabilis (Malpighiaceae) as a visual cue for pollen but not oil foraging by oil-collecting bees. Sci Nat 105(7):1–10
Faegri K, van der Pijl L (1966) The principles of pollination ecology. Toronto and Oxford Press
Gomiz NE, Torretta JP, Aliscioni SS (2013) Comparative anatomy of elaiophores and oil secretion in Gomesa (Orchidaceae). Turk J Bot 37:859–871
Gomiz NE, Torretta JP, Aliscioni SS (2014) Zygostates alleniana (Orchidaceae: Epidendroideae: Cymbidieae: Oncidiinae): estructura floral relacionada a la polinización. An Jard Bot Madr 71:e002
Gomiz NE, Torretta JP, Aliscioni SS (2017) New evidence of floral elaiophores and characterization of the oil flowers in the subtribe Oncidiinae (Orchidaceae). Plant Syst Evol 303:433–449
Inouye DW (1981) The terminology of floral larceny. Ecology 61(5):1251–1253
Johnson SD, Schiestl FP (2016) Floral mimicry. Oxford University Press, Oxford
Maloof JE, Inouye DW (2000) Are nectar robbers cheaters or mutualists? Ecology 81(10):2651–2661
Mangiafico SS (2015) An R Companion for the Handbook of Biological Statistics, version 1.3.2. [Retrieved from rcompanion.org/rcompanion; Accessed 21 Nov 2021]
Martins AC, Melo GAR, Renner SS (2015) Gain and loss of specialization in two oil bee lineages, Centris and Epicharis (Apidae). Evol 69:1835–1844
Martins AC, Melo GAR (2016) The New World oil-collecting bees Centris and Epicharis (Hymenoptera, Apidae): molecular phylogeny and biogeographic history. Zool Scr 45:22–33
Maubecin CC, Rocamundi N, Palombo N, Aguirre LA, Cocucci AA, Sérsic AN (2021) Teasing out the functional groups of oil-collecting bees in the light of the pollination of Nierembergia flowers. Arthropod-Plant Interact. https://doi.org/10.1007/s11829-021-09864-y
Michener CD (2007) The bees of the world, 2nd edn. Johns Hopkins, Baltimore
Moure JS, Melo GAR, Vivallo F (2007) Centridini Cockerell and Cockerell, 1901, in: Moure, J. S., Urban, D. and Melo, G. A. R. (Eds.). Catalogue of bees (Hymenoptera, Apoidea) in the Neotropical region. Sociedade Brasileira de Entomologia, Curitiba, pp. 83–142
Neff JL, Simpson BB (1981) Oil-collecting structures in the Anthophoridae (Hymenoptera): morphology, function, and use in systematics. J Kansas Entomol Soc 54(1):95–123
Neff JL, Simpson BB (2017) Vogel’s great legacy: the oil flower and oil-collecting bee syndrome. Flora 232:104–116
Oliveira LC, Nunes CEP, Brito VLG, Caetano APS (2022) Floral oil production in a family dominated by pollen: the case of Macairea radula (Melastomataceae). Flora 288:152008. https://doi.org/10.1016/j.flora.2022.152008
Oyarzabal M, Clavijo J, Oakley L, Biganzoli F, Tognetti P, Barberis I, Maturo HM, Aragón R, Campanello PI, Prado D, Oesterheld M, León RJC (2018) Unidades de vegetación de la Argentina. Ecol Austral 28:040–063
Proctor M, Yeo P, Lack A (1996) The natural history of pollination. Harper Collins Publishers, London
R Development Core Team (2013) R: a language and environment for statistical computing. (R Foundation for Statistical Computing: Vienna, Austria) [on line] http://www.r-project.org (Accessed on Oct 15 21)
Reis MG, Singer RB, Gonçalves R, Marsaioli AJ (2006) The chemical composition of Phymatidium delicatulum and P. tillandsioides (Orchidaceae) floral oils. Nat Prod Commun 1, 1934578X0600100911
Renner SS, Schaefer H (2010) The evolution and loss of oil-offering flowers: new insights from dated phylogenies for angiosperms and bees. Philos. Trans. R. Soc. London. Ser B 365:423–435
Reposi SD, Gotelli MM, Torretta JP (2021) Anatomy and ultrastructure of floral osmophores of Catasetum fimbriatum (Orchidaceae). Protoplasma 258:1091–1102
Rocha-Filho LD, Garófalo CA (2016) Natural history of Tetrapedia diversipes (Hymenoptera: Apidae) in an Atlantic semideciduous forest remnant surrounded by coffee crops, Coffea arabica (Rubiaceae). Ann Entomol Soc Am 109(2):183–197
Roig-Alsina A (1997) A generic study of the bees of the tribe Tapinotaspidini, with notes on the evolution of their oil-collecting structures (Hymenoptera, Apidae). Mitt Münch Entomol Ges 87:3–21
Roig-Alsina A (1999) Revisión de las abejas colectoras de aceites del género Chalepogenus Holmberg (Hymenoptera, Apidae, Tapinotaspidini). Rev Museo Argent Cs. Nat., n.s. 1(1):67–101
Roig-Alsina A (2000) Claves para las especies argentinas de Centris (Hymenoptera, Apidae), con descripción de nuevas especies y notas sobre distribución. Rev. Museo Argent. Cs. Nat., n.s. 2:171–193
Shorthouse DP (2010) SimpleMappr, an online tool to produce publication-quality point maps. [on line] http://www.simplemappr.net (Accessed 15 Dec 21)
Sigrist MR, Sazima M (2004) Pollination and reproductive biology of twelve species of Neotropical Malpighiaceae: stigma morphology and its implications for the breeding system. Ann Bot 94:33–41
Singer RB, Cocucci AA (1999) Pollination mechanisms in four sympatric southern Brazilian Epidendroideae orchids. Lindleyana 14:47–56
Steiner KE (1985) Functional dioecism in the Malpighiaceae: the breeding system of Spachea membranacea Cuatr. Amer J Bot 72(10):1537–1543
Torretta JP, Marrero HJ, Roig-Alsina A (2011) Chalepogenus roitmani Roig Alsina (Hymenoptera: Apidae: Tapinotaspidini): description of the male and new geographical records for the species. Zootaxa 2797:21–24
Torretta JP, Roig-Alsina A (2016) First report of Monoeca Lepeletier and Serville in Argentina, with description of two new species (Hymenoptera: Apidae). J Melittology 59:1–12
Torretta JP, Roig-Alsina A (2017) Las abejas colectoras de aceite del género Paratetrapedia Moure (Hymenoptera, Apidae, Tapinotaspidini) en la Argentina. Rev. Museo Argent. Cs. Nat., n.s. 19, 131–140
Torretta JP, Aliscioni SS, González-Arzac A, Avalos AA (2017) Is the variation of floral elaiophore size in two species of Stigmaphyllon (Malpighiaceae) dependent of interaction with pollinators? Plant Ecol. Divers 10:403–418
Vilas Boas JC, Fava WS, Laroca S, Sigrist MR (2013) Two sympatric Byrsonima species (Malpighiaceae) differ in phenological and reproductive patterns. Flora 208:360–369
Vinson SB, Frankie GW, Williams HJ (1996) Chemical ecology of bees of the genus Centris (Hymenoptera: Apidae). Fla Entomol 79:109–129
Vinson SB, Williams HJ, Frankie GW, Shrum G (1997) Floral lipid chemistry of Byrsonima crassifolia (Malpighiaceae) and a use of floral lipids by Centris bees (Hymenoptera: Apidae). Biotropica 29:76–83
Vogel S (1974) Ölblumen und ölsammelnde Bienen. Tropische Und Subtropische Pflanzenwelt 7:1–267
Vogel S (1990) History of the Malpighiaceae in the light of pollination ecology. Mem New York Bot Gard 55:130–142
Vogel S, Cocucci AA (1995) Pollination of Basistemon (Scrophulariaceae) by oil-collecting bees in Argentina. Flora 190:353–363
Vogel S, Machado IC (1991) Pollination of four sympatric species of Angelonia (Scrophulariaceae) by oil-collecting bees in NE. Brazil Plant Syst Evol 178(3):153–178
Acknowledgements
We thank to A. Roig-Alsina for collaboration in the determination of bees and critical reading of preliminary version of manuscript, to three anonymous reviewers, to F. Vivallo for determination of Centris proxima, to A. Cocucci and L. Rocha-Filho for bibliographic queries, and to R. Saurral for helping with the English revision. To the family Gotz and Alparamis S. A. for the permission to conduct this study in Reserva El Bagual, and to A. Di Giacomo for logistical support, the Administración de Parque Nacionales (Regional NEA), the Ministerio de Ecología y Recursos Naturales Renovables, province of Misiones, and the Dirección de Áreas Naturales Protegidas, Organismo Provincial para el Desarrollo Sostenible, province of Buenos Aires, for permission to conduct part of this study in protected areas.
Funding
This work was supported for JPT (grant PIP 11220110100312 of Consejo Nacional de Investigaciones Cientıficas y Técnicas (CONICET), and grants UBACyT 20020130200203BA and 20020170200252BA of the Universidad de Buenos Aires.
Author information
Authors and Affiliations
Contributions
JPT conceived this research; HJM performed statistical analysis; JPT wrote the paper; and all authors participated in the field work and revisions, and read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was performed with permission of the Administración de Parque Nacionales (Regional NEA), the Ministerio de Ecología y Recursos Naturales Renovables, province of Misiones, and the Dirección de Áreas Naturales Protegidas, Organismo Provincial para el Desarrollo Sostenible, province of Buenos Aires,
Informed consent
Informed consent was obtained from all individual participants included in the study.
Consent for publication
AAA provided informed consent for publication of all photographic images in Figs. 1 and 2 (except 2c). JPT provided informed consent for the publication of the photographic image in Figure 2c.
Conflict of interest
The authors declare no competing interests.
Additional information
Manuscript editor: James Nieh
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Torretta, J.P., Aliscioni, S.S., Marrero, H.J. et al. Oil flowers of Malpighiaceae and its oil-collecting bees: loyalty and robbery in a highly specialized system. Apidologie 53, 30 (2022). https://doi.org/10.1007/s13592-022-00945-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13592-022-00945-2