Research Article
State-of-knowledge on Cariniana estrellensis (Raddi) Kuntze (Lecythidaceae) for Genetic Conservation in Brazil
Embrapa Genetic Resources and Biotechnology, Parque Estayao Biol6gica-PqEB, CP 02372, 70770-900 Brasilia, DF, Brazil
Conservation operating through in situ channels has, understandably, been ecosystem focused. There is, however, need to complement this with attention directed at particular target species. Here, this approach is developed for Cariniana estrellensis (Raddi) Kuntze, a forest species, which is typical of a distinctive but threatened habitat in central Brazil, the Cerrado, although not necessarily restricted to it. This taxon is included in the listing of 23 priority species for Brazil drawn up by the Brazilian Agricultural Research Organisation through its Genetic Resources and Biotechnology Research National Centre (Embrapa-Cenargen, 1988) for the Brazilian Government in connection with the national conservation strategy. Criteria for selection were those recommended by the FAO Panel of Experts on Forest Gene Resources: restricted ecological distribution, low population densities, heavy exploitation for wood and other products, unknown or deficient silviculture and threatened habitat (Roche, 1987).
Efforts to trace a robust but flexible standard system for acquiring, collating and organising information on named neotropical tree species were not successful. The reasons always reside in problems of standardisation of taxonomic and systematic nomenclature and regional peculiarities of (and particularly for wide-ranging) woody species, as well as the generally scattered nature of the acquired knowledge on these species. For example, in the Forestry compendium of CAB International (2000) C. estrellensis is only briefly treated as an outline data sheet. However, Lorenzi (2000) provides an instructive account, including figures, of this species. Reference to World Conservation Monitoring Centre (WCMC-Kew, 1990) indicated that for other tropical areas the situation was no different. Since species are the targets of gene resource conservation, a structured review of existing knowledge has great appeal when conservation strategies are to be formulated. Accordingly, this study offers a framework for such action, with C. estrellensis serving as a case study.
The form and distribution of knowledge about tropical tree species introduce complications in information retrieval. Un-processed raw data (for example, those recovered from herbarium holdings, ecological literature and species listings) need to be especially reviewed and critically incorporated into the progressing study. In an uncollated state, published information is often very dispersed, much (notably early references) being in obscure or poorly circulated documents. Various languages may be involved and reference may be made under obsolete names, which may reflect broader or narrower concepts of the species than those accepted today. For many wide-ranging species these problems are acute, making the preparation of a unifying account taxing, time-consuming and sometimes laborious. The exercise is nevertheless important as the basis of an authoritative conservation plan.
Few can claim familiarity with all aspects of the existing knowledge of a wide-ranging species: existing information about a species can be allocated in many fields, each the line of a different group of specialists: foresters, ecologists, geographers, botanists. Geographical spread complicates matters greatly. Much concern with species is at national rather than range-wide level. Differences in the circumscription and in the nomenclature of species need to be recognised (and perhaps reconciled) and taken into account as the information is integrated. Whether or not this can be achieved in a single step depends on the quantity of published and archival information (including herbarium material), on the species, as well as on the resources and time available to the investigator.
Aimed at producing a current state-of-knowledge account for the species to support genetic conservation programmes in Brazil, the present study has three objectives: (1) to review available information on the biology, ecology, silviculture and management of C. estrellensis relevant to its genetic conservation as a sustainable resource; (2) to create a comprehensive and authoritative monographic account of the species by a critical analysis of available information, highlighting in the process unresolved misinterpretations and inconsistencies which need clarification and (3) to identify the major gaps in current knowledge of the taxon and recommend positive research actions to fill them.
This study is devoted to an in-depth account of the target taxon included in the programme of conservation of forest genetic resources in Brazil, which is typical of the Central Brazilian Cerrado region. Consequently, it has copious monographic content for the species. Comprehensiveness and authority have been sought by means of the extensive, though scattered information accessed and the scope of the review has enabled a clear picture of the species throughout the neotropics. Comprehensive comments on taxonomic and morphological aspects-at times addressing exposed contradictions and misinterpretations-are given, along with résumés of what is known of the reproductive biology and associated phenology. Distribution and ecology are aspects dealt within the context of the overall taxon range. Silvicultural and management experience was revealed but the information retrieved was inconsistent, scattered, incomplete and seldom widely circulated. As expected utilisation of the resource was particularly related to wood properties, but a variety of comments on alternative non-timber usage for the species were located. Lastly, information on genetic conservation status and priorities was supplied as general introductory statements, which were linked to conservation action to be taken and discuss later.
The approach followed has been adapted from that used by Hall and Walker (1991) for Balanites aegyptiaca and the schemes of Mondal (1986) for Maesopsis eminii and Irozuru (1986) for Nauclea diderrichii and the approach followed by Morse (1981) to report the conservation status of an endangered North American shrub species.
The species is considered sequentially with a subdivision allocated for each main aspect. The first subdivision outlines the systematic position and circumscription of the taxon. The nomenclatural history in each case is presented as a tabular chronological summary. The second subdivision is descriptive, covering, in turn, the seedling phase and the mature tree with, for the latter, details of habit, size and form, foliage, flowers and fruits and, where known, systematic anatomy. The third subdivision is in three sections covering the reproductive biology of the taxon with respect to phenology, pollination and dispersal and seed biology in natural conditions. The fourth subdivision is in two sections: distribution and ecology. The distribution covers, as far as possible the whole natural range. The ecology is treated first by factor: elevation, climate and soils and then in the phytosociological context (associated species, relations with the natural fauna and with forest disturbance). The 5th subdivision addresses the silviculture and management of the species. In terms of artificial regeneration, experience involving propagation and outplanting and establishment and the performance of planted individuals is reported. On the management side, an account of attention in forest stands is given. The sixth subdivision deals with utilisation. There are three subsections-a description of the wood properties regarding macro- and microscopic, as well as physical and mechanical features; use as timber in terms of seasoning and preservation, durability and workability and final uses; other uses, based on the Booth and Wickens (1988) scheme of non-timber uses for African species. The seventh and final subdivision is concerned with the conservation status of the species.
The fragmented and unsatisfactory species information for many species in the neotropics requires today’s monographers to invest more thought, effort and time to taxonomic circumscriptions and their geographical implications than their counterparts in Africa, Asia or Australia.
In the present research, a retrospective picture of the taxonomic chronology was a necessary preliminary to an overview of the knowledge base.
More specific comments on the importance of knowledge of the different aspects of a species have been given by Hall (1993 and 1994). Hall reports the general need for monographic accounts rather than only lists, brief profiles, data-sheets and bibliographies on both multipurpose tree species and traditional forestry trees. Monographs can be the most authoritative and comprehensive documents of the state of knowledge on any species. However, it is important that they are based as a sound framework in the form of a set of headings covering all aspects of the tree’s biology and resource potential: taxonomy/morphology, distribution, ecology and silviculture/management. Critical assessment of the information gathered is important, especially resolving or explaining nomenclatural contradictions and differences of opinion as far as possible. Hall (1993 and 1994) stress the prominent role distribution maps should play among those headings. This is an exercise, which depends on information from five distinct sources-herbaria, taxonomic literature (basically in the form of floras), inventories, ecological literature and personal observations. Complementary data sets can cover terrain, climate and soil allowing occurrences to be related to these factors. Hall (1994) also discusses the potential combined role of such information sets in distinguishing genetically determined variation from ecologically determined variation in a wide-ranging species once mapped using all relevant available information. A critical approach to these issues is essential if conservation programmes are to be successful.
Unification of information on a species into a structured and authoritative monograph has been achieved for the African dry zone tree B. aegyptiaca (L.) Delile (Balanitaceae) by Hall and Walker (1991). This study serves as a useful model for a monograph but adaptations will be appropriate for other species. In the present case, prominence has been given to the conservation aspect, which has been excluded by Hall and Walker (1991) although separately considered at some length elsewhere (Hall, 1992). This conservation aspect is also covered in more recent monographs such as those presented by Hall et al. (2000) for Prunus africana and Pasiecznik et al. (2001) in a Prosopis juliflora-Prosopis pallida complex account. The present study involves diverse sources of information. For Brazil these are published and unpublished material at Embrapa and Cenargen, National Parks management plans and lists and maps of current protected areas at the Brazilian Institute for the Environment and Renewable Natural Resources-Ibama. Paraguayan Conservation Data Centre data sheets on the target taxa were also obtained. In Britain, WCMC’s database information on protected areas and assessment of conservation status of the target species were consulted and information assembled through visits to and correspondence with the Royal Botanic Garden, Kew (both herbarium and library). Electronic (Web of Science, CABI TreeCD) and traditional abstracting facilities (Forestry and Biological Abstracts) and a range of periodical publications but particularly Forest Ecology and Management, Forest Genetic Resources Information, Threatened Plants Newsletter and Silvae Genetica were also consulted. For less readily available recent documents Inter Library loans were used. Further information was collected in discussions and correspondence with experts-both in Britain and abroad (Brazil, Paraguay, Switzerland). This review has been done as complementary to field studies carried out with four target species occurring in the gallery forests of the central Brazilian Cerrado-a savanna-like vegetation physiognomy- in the early nineties.
Vernacular Names of Cariniana estrelensis
Portuguese: jequitibá-rosa, jequitibá, jequitibá-rei, jequitibá-branco, jequitibá-vermelho, estopeira, estopa, bingueiro, pau-caixão, coatinga, pau-carga, pau-estopa, cachimbeiro (Occhioni, 1975; Prance and Mori, 1979; López, 1987; Salomão, 1990).
Spanish: mussambe (Prance and Mori, 1979).
Guarany: kai kay’gua (López, 1987).
Systematic Position and Circumscription
The taxonomic history, including synonymy, of C. estrellensis is shown in Table 1.
Although during the present century several species have been added to Cariniana, the circumscription of the genus has not changed since its reinstatement by Miers, in 1874, according to Prance and Mori (1979). The same authors reported that Knuth, in 1939, listed 14 species but one was Couratari paraensis and misplaced. Since Knuth’s review two species have been added to the genus (Prance and Mori, 1979). The species of Cariniana currently accepted are:
C. decandra Ducke
C. domestica (Martius) Miers
C. estrellensis (Raddi) Kuntze
C. ianeirensis R. Knuth
C. integrifolia Ducke
C. kuhlmannii Ducke
C. legalis (Martius) Kuntze
C. micrantha Ducke
C. multiflora Ducke
C. pachyantha A. C. Smith
C. pauciramosa W. Rodrigues
C. penduliflora Prance
C. pyriformis Miers
C. rubra Gardner ex Miers
C. uaupensis (Spruce ex Berg) Miers
| Table 1: | Taxonomic history: Cariniana estrellensis (Raddi) Kuntze |
 | |
DESCRIPTION
Seedling
Barbosa (1984) describes the germination of C. estrellensis and C. legalis endorsing Prance and Mori’s (1979) observation of the cotyledons being withdrawn from the seed coat, characterizing phanerocotylar cotyledons, a feature of all neotropical Lecythidaceae with foliaceous cotyledons.
The integument is reported to break in ca 10-15 days, through the micropylar region, with consequent appearance of the radicle. The hypocotyl elongates within two to four days of germination. About ten days after germination, the radicle is ca 7.5 cm long and the hypocotyl about 2.0 cm long. The seed coat (testa) is lifted with the cotyledons and shed a few days later. The leaf-like cotyledons are green, cordate, palmately veined and large and vigorous. The epicotyl starts development about one month after germination begins. At this stage the hypocotyl is ca 4.5 to 5.0 cm and the cotyledons are still present and turning reddish. At the beginning of the development of the epicotyl, a tiny stipule emerges. Pubescence extends to the point of insertion of the first eophyll. These first leaves (eophylls) are alternate, serrately margined and larger than the leaves of adult individuals. They have expanded 40 days after germination, when the radicular system is already well developed. The cotyledons start falling after 6 months when fall of the old eophylls and emergence of new leaves also occurs (Barbosa, 1984).
Mature Tree
Habit, Size and Form
Prance and Mori (1979) refer to the growth form of Cariniana as leptocaul. The authors reckon that this is an oversimplified description of the form which corresponds to at least five of the architectural types described by Hallé and Oldeman (1970) and Hallé et al. (1978).
It is a straight, long - but can also fork (López, 1987) - cylindrical trunk with enlarged base (Occhioni, 1975; López, 1987), but also sometimes presenting straight base (Barbosa, 1984). The crown is rather large and wide, straight or inversely conic to rounded in shape. Thick alternate plagiotropic branches are inserted at ca 30° on the trunk and are often covered with epiphytes and carry fairly light to dense foliage (Occhioni, 1975; Barbosa, 1984; López, 1987). When young, the branchlets are sparsely puberulous but soon become glabrous (Prance and Mori, 1979). Lenticels are present throughout the branchlets (Barbosa, 1984).
The tree can be as small as 20 mx60 cm in height and trunk diameter, respectively (Barbosa, 1984), but reaches 25-38 mx80-250 cm in Paraguay (López, 1987) or 40-45 mx300-400 cm (Occhioni, 1975) in Brazil, or 37 m tall (Miers, 1874 reported by Record and Hess, 1972), or even 50 m in height and 500 cm in trunk diameter (Salomão, 1990). It should be noted, however, that such huge diameters reflect measurement at the base, which is normally expanded (Occhioni, 1975)-cf (Fig. 1). A straight cylindrical bole is reported, reaching a length of 8-18 m (López, 1987), 15-18 m (Barbosa, 1984), 12-22 mx60-150 cm diameter (Salomão, 1990) and 25-30 mx400+ cm diameter (Occhioni, 1975). The outer bark is grooved, rough, fibrous and reddish-grey to dark-grayish in colour with longitudinal furrows 10-16 cm long, 2-3 cm wide. It is rather thin - less than 1 cm - whilst the inner bark is quite thick - up to 4.5 cm -, uniform, whitish, slightly pinkish to yellowish, exuding a limited amount of greenish white latex when cut. Between the two layers of bark there is a thin reddish layer which is not uniform, but rather irregular following the contour of the outer bark (Occhioni, 1975; Barbosa, 1984; López, 1987; Salomão, 1990).
Foliage
The leaves are simple, alternate and petiolate, with the lamina oblong-elliptic in shape, 3-11 cm long (7-10 cm according to Occhioni, 1975; 4-6 cm according to Barbosa, 1984) and 1.5-6.0 cm broad (2.0-3.5 cm according to Barbosa, 1984). The margins are serrate and the apex acuminate, the acumen being 2-5 mm long and slightly curved. The leaf base is subcuneate and decurrent on the petiole. The midrib is flat above, prominent beneath and glabrous (or slightly puberulous beneath). The venation is pinnate (camptodromous-brochidodromous), with the primary veins in 11-18 pairs, lacking domatia at the junctions with the midrib. These veins are 2-5 mm apart at base, prominulous above, prominent beneath and anastomose near the margin. There are some 12 pairs of conspicuous secondary veins (Occhioni, 1975; Barbosa, 1984). The petiole is short - 5-13 mm long - and slightly winged (Prance and Mori, 1979; López, 1987).
Flowers and Fruits
The inflorescences (Fig. 1) are axillary racemes or racemose panicles 3-6 cm long arising directly from the branches. The rachis is puberulous. The pedicels are 2-5 mm long, slender and puberulous. The bracts and bracteoles are small and caducous. There is an articulation where the pedicel is attached to the rachis. The bracteoles are inserted directly below the attachment. Occhioni (1975) noted few flowers (5-12) in the inflorescence but López (1987) draws attention to the presence of numerous flowers. The calyx is 1.0-1.5 mm long and campanulate with six broadly triangular lobes, each sparsely puberulous externally. The corolla is of six obovate petals each 4-7 mm long and creamish-white in colour (Occhioni, 1975; Prance and Mori, 1979). The androecium, is only prolonged on one side, characterizing a zygomorphic flower (although regarded by Prance and Mori, 1979 as an actinomorphic state because the zygomorphy is so slight compared to other genera in the Lecythidaceae). It is ca 5 cm in basal diameter and white in colour, with about 40 stamens, inserted all over the interior but more densely towards the apex. The ovary is inferior (Barbosa, 1984) and tri-locular. The style is very short (Fig. 2).
The fruit is a cylindrical-oblong brown pyxidium 6.5-9.5 cm long, 3.0-3.5 cm broad, the calcyne ring is ca 1 cm below apex, the pericarp is thin (ca 1 mm thick at the apex although thicker below) and there is a row of tooth-like protrusions around the line of opercular dehiscence. The operculum is 1.5-2.5 cm in diameter at the apex and up to 7 cm long. The columella is triangular. The seed wings are up to 3.5 cm long and 1.2 cm broad. There are (5) 6 (17) seeds (Barbosa, 1984) - more or less pyriform in shape, each 1.2 cm long and 0.6 cm broad (Salomão, 1990).
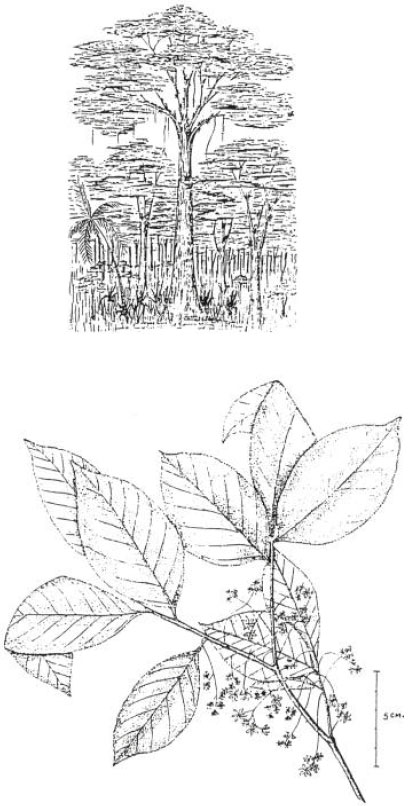 | |
| Fig. 1: | Habit and form of C. estrellensis and branchlet with inflorescence (Occhioni, 1975) |
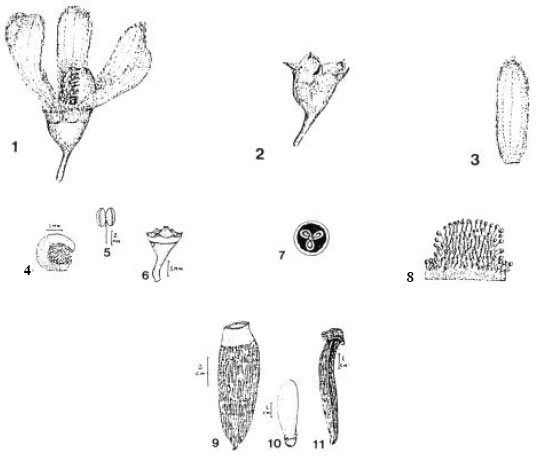 | |
| Fig. 2: | Cariniana estrellensis flower complete, 1 (x4.3) and without petals and androecium, 2 (x4.3); petal, 3 (x4.3) androphore, 4; stamen, 5; ovary, 6; cross section of ovary, 7 (x4.3) androecium opened up, 8 (x4.3); fruit, 9; seed, 10; operculum, 11 (modified from Occhioni, 1975 and Prance and Mori, 1979) |
The conspicuous features of the embryo are the leaf-like cotyledons, the long radicle and the unilateral wings - an aril flattened out (Occhioni, 1975; Prance and Mori, 1979; López, 1987).
Systematic Anatomy
Prance and Mori (1979), with scanning electron microscopy, revealed minute features of the leaf surface confirming the absence of hairs (Fig. 3). The cuticle is more or less smooth and the stomata are often sunk within stomatal crypts and/or surrounded by papillae - features likely to reduce transpiration losses.
Reproductive Biology
Phenology
The flowering time varies with locality. Table 2 shows phenological information available for different parts of the species range.
Although Prance and Mori (1979) indicate for the Lecythidaceae a general pattern of flowering during the dry season this seems not to be the case for this particular species.
Pollination and Dispersal
Allogamy predominates in the family (Barbosa, 1984). Prance and Mori (1979) note a lack of information regarding pollination in ‘actinomorphic’-flowered genera, with the exception of Gustavia.
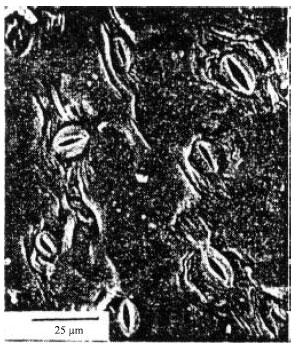 | |
| Fig. 3: | SEM photomicrography of the abaxial leaf surface of C. estrellensis showing stomates in crypt (Prance and Mori, 1979) |
| Table 2: | Phenology of Cariniana estrellensis |
 | |
| Occhioni (1975), Rizzini (1978), Barbosa (1984), López (1987), Cavallari and Gripp (1990) and Salomão (1990) | |
They nevertheless report many pollen-gathering bees (Melipona and Trigona) visiting flowers of Cariniana legalis a close relative of C. estrellensis. Pollen seems the only reward to these visitors: no nectar is produced.
Barbosa (1984) observed pollination on C. estrellensis and reported that the slightly scented flowers open before dawn. The anthers, which are closed, liberate the pollen only after the opening of the flower. The visiting intensifies one hour after sunrise. The androecium and petals fall together in the afternoon. There is no mass production of flowers, which are well distributed over the crown. There is a physical barrier partially protecting pollen and stigma from contact with the insect having to get into the urceolus to touch the stigma.
The unilateral winged seeds of this canopy species are dispersed by anemochory. Prance and Mori (1979) point out two features favour dispersal by wind: the aril flattened out to form a wing and the emergent position in riverine habitats of most Cariniana species, enabling exposure of the fruits to maximum wind velocities.
Barbosa (1984) reports that, on opening, the mature fruit loses as a single entity, the woody columella and the operculum. The seeds are released in the wind, while the fruit remains on the branches, dropping eventually and staying under the tree for long periods (López, 1987).
Seed Biology in Natural Conditions
Instituto Florestal (1978) reports abundant seed production but heavy depletion by monkeys. There are 9,090 to 12,195 seeds to the kilogram, extrapolating data from Salomão (1990).
Distribution and Ecology
Distribution
Present Distribution
The species occurs (Fig. 4) in Brazil, Bolivia and Paraguay (Prance and Mori, 1979; López, 1987). More recently, data were also gathered for Peru, using the W3 Tropicos Database of Missouri Botanic Gardens.
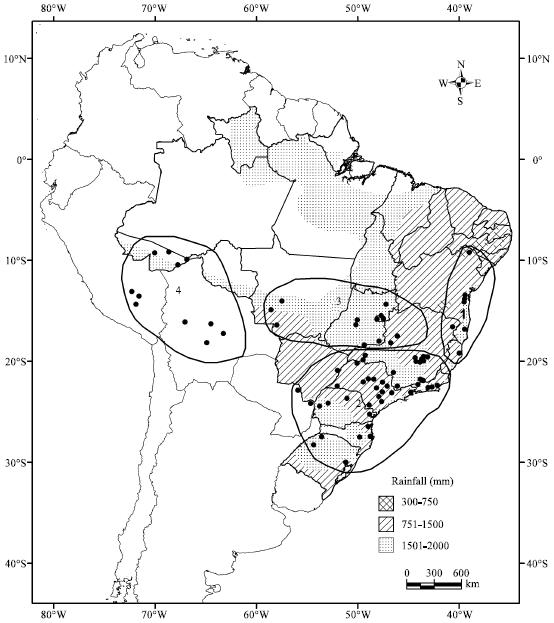 | |
| Fig. 4: | Distribution of C. estrellensis in S. America. 1, northeastern Brazil; 2, southeastern and southern Brazil; 3, central Brazil; 4, western Brazil, Bolivia and Peru |
Range
Cariniana estrellensis is the only species in the family which has been collected south of the Tropic of Capricorn. It is mainly found in the rain forests on the coast of Brazil from southern Bahia to Rio Grande do Sul states1,2 but it is also quite common in the central Brazilian Cerrado3, occurring in the riverine forests, where it is a common dominant tree species. It occurs also inland in the states of São Paulo and Minas Gerais2 in even drier forests. This centre-south distribution in Brazil constitutes the core of the range. It is present in the State of Acre4, this being the northernmost recorded presence of the species and the Amazonian marginal boundary of its geographical distribution. Further east, the northern limit is in the northern part of the State of Bahia1. Outside Brazil the species is reported from the northeastern parts of Bolivia and Peru4 and Paraguay2 - forests in the north of the Oriental region (López, 1987). The western and northern occurrences seem to represent disjunct populations, but Brown Jr. and Prance (1987) have suggested species populations in central Brazil Cerrado gallery forests as links between the Amazonian and the Atlantic coast forests.
Ecology
Environment
Elevation
Although Prance and Mori (1979) regard the Lecythidaceae a lowland family, reporting only Eschweilera spp., Gustavia spp. and Lecythis karavaiensis Steyerm. to occur above 1,000 m altitude, C. estrellensis should, however, be included on this list. The presence of the species in the Brasília region (1,100 m) is an example of this alongside the herbarium holdings examined for other parts of the range.
Soils
Although López (1987) reports deep clay soils as a preference of the species in Paraguay, it is quite certain these occurrences on richer luvisols are not as representative as the prominence of the species on poorer soils in the centre-south Brazilian territory. Indeed, prominence on luvisols in the northeast of Brazil is also low.
Climate
Comments relate to a detailed mean annual rainfall map of South America (WMO et al., 1975) and association with the distribution map (Fig. 4).
The range of mean annual precipitation for C. estrellensis is mostly between 1,200 and 1,600 mm. Departures from these are only observed in outlying occurrences. In northern parts of the range precipitation reaches up to 2,000 mm but in western sub-Andean occurrences precipitation decreases to 800 mm and, in northeastern Brazil, even lower amounts (600 mm) are observed.
Site
As with other Lecythidaceae, this is a species of mesic rather than wet or dry habitats. This could explain prominence of the species in fairly wet forests although it is deciduous. The low representation of the family in drier habitats reflects the inability of most species to form mycorrhizal associations as when compared with Leguminosae, which is more frequent in such habitats due to the capability of associating with mycorrhyza (Prance and Mori, 1979). López (1987) reports the presence of C. estrellensis in the uppermost stratum of tall forests: it is one of the largest trees in such sites. Barbosa (1984) indicates such a status in the gallery forests in the Cerrado. Adult individuals are almost always emergent. Rizzini (1978) reported low frequency: 2 trees ha-1 and a volume of 22 m3 ha-1 in the coastal forest of Bahia State. However, the volume varies greatly: Heinsdijk et al. (1965), cited by Rizzini (1978), found in nearby northern Espírito Santo only 7 m3 ha-1.
Cariniana estrellensis in a Phytosociological Context
Associated Species
Table 3 shows species associated with C. estrellensis.
| Table 3: | Associated woody species of Cariniana estrellensis in three phytogeographic provinces in a number (*) of sampled areas. AF (Atlantic coast forest), RF (southeastern Brazilian rainforest), C (Cerrado) |
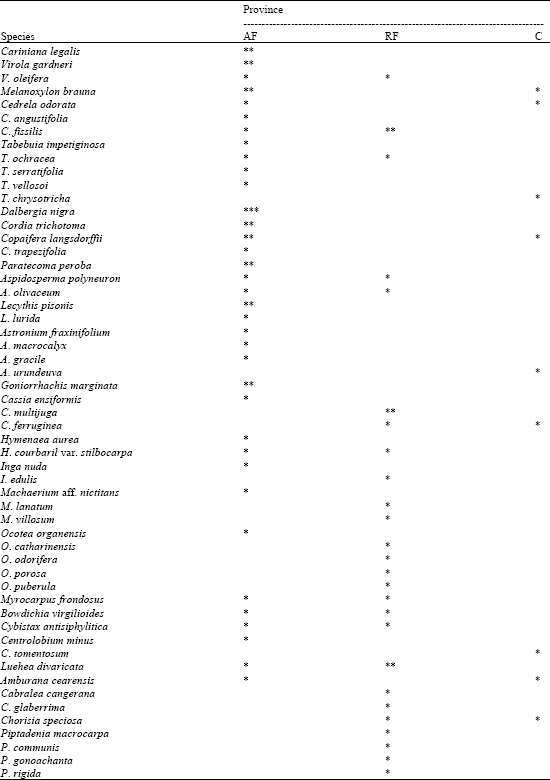 | |
| Source: Salomão (1990) | |
Relations with the Natural Fauna
Instituto Florestal (1978) reported heavy depletion of the massive seed production by monkeys.
Relations with Forest Disturbance
Prance and Mori (1979) drew attention to the fact that Lecythidaceae, this species included, are usually absent from secondary vegetation, especially if the forest is burnt after the felling.
Silviculture and Management
Experience with Artificial Regeneration
Propagation
Species of Lecythidaceae, including C. estrellensis, are reported to sprout from cut stumps when there is no burning after felling the vegetation (Prance and Mori, 1979). Artificial regeneration of the species is mainly by seed. Barbosa (1984) describes seed collection and handling preparatory to silvicultural trials and enrichment planting. Seed is sought from healthy and vigorous trees, with no deformities. The size reached by mature individuals necessitates use of special collection gear (spikes, aluminium ladders, bicycles or ropes). After collection, fruits are left to dry out until they open and the seeds are then extracted. Storage, if necessary, follows de-winging.
A fairly short germination period is reported: 10-15 days (Barbosa, 1984), 15-20 days (Instituto Florestal, 1978; López, 1987; Salomão, 1990). Barbosa (1984) reports very low-about 1%-germination percentages under laboratory conditions for two provenances: apparently normal seeds mainly contained rudimentary embryos or were empty.
Barbosa (1984) advises direct sowing in plastic bags to simplify subsequent operations. Three seeds are sown per bag on a well-watered, moist medium. Seeds are covered with a thin layer of sieved soil. The selection of the seedling for retention is undertaken at the two-leaf stage - the rejects are culled.
Outplanting and Establishment
Seedlings are drought-hardened when they reach 25-30 cm in height by having irrigation reduced. Attainment of a height of 25 cm approximates to the onset of a phase of accelerated growth (Barbosa, 1984).
Performance of Planted Individuals
In Brazil, C. estrellensis is cultivated on an experimental basis in two places in Minas Gerais State and in the states of Bahia and Rio de Janeiro. Outside Brazil it is grown in Zimbabwe (Prance and Mori, 1979).
For 43 trees aged 16 years, in Rio de Janeiro, Almeida (1947) cited by Rizzini (1978) observed a maximum height of 20 m, a minimum height of 8 m, a maximum diameter of 30 cm and a minimum diameter of 12 cm. Most individuals were from 10.5 to 14.6 m in height and from 14 to 20 cm in diameter. For 22 trees 8 years old Almeida reported heights from 3 to 9 m and diameters from 2 to 12 cm.
Barbosa (1984) reports that in a plantation where C. estrellensis constituted the undergrowth in a Pinus elliottii stand individuals had reached an average height of 3.84 m and an average dbh of 2.83 cm at eight years.
In an interesting enrichment planting exercise in the State of São Paulo, Nogueira (1977), cited by Barbosa (1984), reported a commercial height of about 12-15 m at 20 years for the species. In this stand there had been good natural pruning, the boles were straight and the first fruiting had taken place at an age of about 10 years.
López (1987) adds that Brazilian forest researchers had reported growth of 0.6-1.0 m year-1 in height and 0.9-1.2 cm year-1 in diameter in planted stands.
Management Attention in Forest Stands
There is virtually no information available on this aspect for plantations, reflecting the paucity of enrichment and commercial stands.
Utilization
Wood Properties
Macro- and Microscopic Features
The colour of the heartwood varies from yellowish-white to slightly pinkish (Barbosa, 1984), yellowish (Record and Hess, 1972), pinkish to pinkish-brown (López, 1987) to reddish (Occhioni, 1975) or reddish-brown (Record and Hess, 1972) or even purplish-brown (Chudnoff, 1984). Sometimes there are dark streaks within the heartwood which usually is not sharply demarcated from the sapwood. The sapwood varies from yellowish to slightly pinkish (Barbosa, 1984), to pale brown in colour (Record and Hess, 1972; Chudnoff, 1984). Cut longitudinal surfaces are smooth but not shiny, with medium lustre and fine to medium texture (Record and Hess, 1972; Barbosa, 1984; Chudnoff, 1984; López, 1987). No distinctive odour or taste has been detected (Record and Hess, 1972; Barbosa, 1984; Chudnoff, 1984). The grain is straight to interlocked (Record and Hess, 1972; Chudnoff, 1984).
Physical and Mechanical Features
The wood is considered firm, tough, hard and rather light to moderately heavy (497 to 737 kg m-3).
Mechanical properties are summarized in Table 4.
Chudnoff also reported an Air-dry Janka side hardness (resistance to penetration) of 4537 N and an Amsler air-dry toughness (resistance to impact) of 2 kJ.
Use as Timber
Seasoning and Preservation
The timber air dries quickly with only slight tendency to warp or check. American kiln schedules are suggested for 4/4 (10x10 cm) and 8/4 (20x10 cm) stocks. Shrinkage from the green to oven-dry state is as follows: radial 2.8%, tangential 5.4%, volumetric 9.0%. The wood is reported to have good dimensional stability after manufacture (Record and Hess, 1972; Chudnoff, 1984).
Chudnoff (1984) reports that the heartwood is extremely resistant to preservatives although the sapwood is permeable.
Durability and Workability
López (1987) and others have stressed the durability of this timber, especially the decay resistance of deeply coloured specimens. Chudnoff (1984) reports good resistance to dry-wood termite attack.
Chudnoff (1984) noted generally satisfactory working properties, but drew attention to the slight blunting effect on cutting edges. Several species in the genus Cariniana have silica in excess of 0.05% which causes rapid dulling of cutters, although veneers cut without difficulty. According to Record and Hess (1972) it is easy to cut but sometimes difficult to saw.
| Table 4: | Mechanical properties of Cariniana sp. |
 | |
| Chudnoff (1984) | |
Uses
This timber is reported to substitute the use of cedar in Brazil (Occhioni, 1975; Barbosa, 1984). The wood is widely used in Brazil for general construction purposes and carpentry (Record and Hess, 1972; Chudnoff, 1984; López, 1987) but notably for interior parts such as door and window frames, ceiling (Occhioni, 1975), windows and blinds. Barbosa (1984) lists uses as furniture components, cabinet making, boxes and veneer for plywood. Use for shipbuilding, canoes, flooring and turnery is reported by Chudnoff (1984). Record and Hess (1972) cite use in making sugar cases in Rio de Janeiro.
López (1987) reported non-utilization of the timber in Paraguay.
Other Uses
Medicinal
The bark is astringent and used to treat diarrhoea, leucorrhoea and angina (Barbosa, 1984).
Conservation Status
Present Conservation Status
The species is not included in the IUCN Red Data Book but is listed as threatened by the Paraguayan Conservation Data Centre (D.A. Cáceres, personal communication, 1992). Although several protected areas in Brazil contain the species, these areas are individually quite small and the populations are questionably safeguarded against genetic erosion and inbreeding. Populations at the margin of the range do not fall within conservation areas and seem exposed to greater risk. The initiative of M.G. Archer to afforest the Tijuca hills in Rio de Janeiro in 1862 (Barbosa, 1984) is worth mentioning. C. estrellensis was among the species planted. Unfortunately this example has been not followed in the last 130 years, apart from the instance of heterogeneous enrichment planting carried out by J.C.B. Nogueira-in 1977.
Present Conservation Priorities
Cariniana estrellensis is listed by the FAO Panel of Experts as a target species for genetic conservation (Roche, 1987). The FAO Panel of Experts continues to list C. estrellensis as high, global, regional and/or national priority species for programme conservation of forest genetic resources (FAO, 2002). Cenargen includes the species among its priorities for action to conserve intraspecific genetic variability.
This study deals with the conservation issue seen from a species-oriented viewpoint rather than the more commonly seen community-centred strategies. The approach followed draws on current conservation theory adapted to the circumstances peculiar to the reviewed taxon. Biological and genetic conservation frameworks for the species are then presented together with suggestions of priority conservation action.
Success in conserving the genetic variation of widely-distributed taxa depends upon sampling enough of the overall genetic diversity inherent to such taxa. This means that the protected populations must include a sufficient spectrum of genetic variability and therefore also an adequate spectrum of ecological variability. The importance of clinal variation, common in wide-ranging tropical species, has become increasingly understood through comparative provenance trials (Roche, 1984). For species-oriented programmes of genetic conservation it is essential that intraspecific genetic variation takes precedence over interspecific variation (Roche, 1984).
For successful implementation of genetic conservation, there must be a clear strategy reflecting the character of the species. Tropical woody species, such as C. estrellensis have long life cycles, are normally allogamous, have large individuals and mostly survive in a wild state. Consequently, relevant objectives for the strategy are refinement of biological knowledge and development of capability to introduce and breed the species where and as required. Such objectives also define which method of conservation suits a given species and, theoretically, dictates the size of population that must be conserved.
In view of our chronic lack of knowledge of many aspects of tropical species biology and ecology, in situ conservation measures merit the greatest investment of effort. This is because the in situ approach provides for maintenance of dynamic genetic variability more effectively than ex situ. Whilst a level of management intervention may be needed to stabilize populations, our ability to achieve this is usually greater than our ability to create ex situ stands containing an adequate representation of the gene pool.
The appeal of ex situ conservation in recent years has been heavily based on theoretical considerations which do not apply for most tropical high forest species. Implementation of this approach appears most practicable for species which are not part of climax forests; this includes species of more open tropical formations and particularly those which are aggressive, light demanding and widely distributed (e.g., Balanites aegyptiaca-Hall, 1992). For the majority of species from the most humid tropical environments, however, very little has been achieved on the development of reliable methods of storage because of seed recalcitrance (FAO, 1990). Additionally, lack of research restricts our understanding of the extent to which seed storage is a safe method of conserving tropical woody species (Roche, 1975). Alternative approaches such as tissue culture remain unproven as realistic practical methods facilitating ex situ conservation.
Efforts have been made to link key genetic conservation research areas presented by Roche (1975) and National Research Council (1991) with knowledge on the target taxon. Gaps in our knowledge, such as those revealed in this study, were given particular attention. Despite these gaps, the available information can be used as a foundation for offering practical suggestions on potentially rewarding conservation approaches, following Hall’s (1992) model for Balanites aegyptiaca.
A good deal is known about the wood properties, taxonomy and morphology of C. estrellensis. The available information about the distribution of the species could be used as a basis for locating potential in situ conservation areas or for planning germplasm collections for provenance trials. However, to pinpoint precise locations to conserve the genetic variability of this taxon requires further refining of the maps; this must be based on visits to previous collection points as well as to new areas, in order to assess more fully the level of infraspecific variation. Our ecological knowledge about the species, despite the relatively high number of sources, is inconsistent and incomplete. Present information on reproductive biology of the taxon is also unsatisfactory.
Comparison of the distribution of the taxa with major soil units was made using the FAO-UNESCO (1971) soil map. Prominence of C. estrellensis is associated with poor soils in gentle landscapes.
Information concerning artificial regeneration and the influence of management on the species is poor.
This is also true for the conservation status, since information is very limited. There are for Cariniana estrellensis some comments on the extent to which it is threatened, on the South American national Conservation Data Centres’ lists. More noteworthy, however, is the consistent association with several other target taxa on the FAO Panel of Experts on Forest Gene Resources list (Roche, 1987). This was the basic document used by Cenargen to choose target species for the national programme of genetic conservation in Brazil. The main priorities of this programme are the establishment of genetic reserves wherever possible and particularly in existing protected areas. Additionally, ex situ conservation is sought through setting up germplasm banks and provenance and progeny trials. in situ genetic reserves should, however, be as large as possible to alleviate problems of genetic drift and consequent depression of genetic variability that can be expected in the existing small reserves. Both in situ and ex situ methods require management.
In summary, despite our limited knowledge about C. estrellensis, we conclude that is typical of many thinly dispersed, outcrossing, tropical forest trees which are at risk as forest disturbance levels increase and individuals become increasingly isolated.
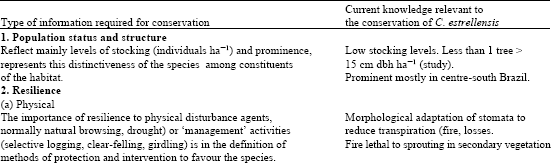
The types of information needed to develop a conservation strategy are presented in Table 5, together with the appropriate data for C. estrellensis.
Although this account deals with the whole natural range of the species, including Bolivia, Brazil, Paraguay and Peru, emphasis, on this occasion, is given exclusively to Brazil, for this was the primary objective of the study regarding assessment of the resource in protected areas and further actions to conserve it.
Of the recognized protected areas (C. Sharpe, WCMC, personal communication, 1992) in Brazil, theoretical conservation suggests few qualify as effective both in size and legal status for the long term conservation goals of forest genetic resources. A more practical listing of potentially viable protected areas is one according with Ibama’s definition (J.C. Gonchorosky, personal communication, 1992) of conservation units of indirect use (national parks, biological reserves, ecological stations) and direct use (national forests and extractive reserves).
Consideration of the species and its distribution within Brazil in relation to the WCMC map of protected areas (C. Sharpe, personal communication, 1992) can be done only on a very tentative basis at present. Nevertheless, it is appropriate to associate occurrences of the taxon with the protected areas to the extent possible.
This was done by showing the distribution of protected areas which have had some more attention regarding their floristic composition, enabling, therefore, to have a picture of the occurrence of the species in such areas (IBGE, 1993). Figure 5 shows a map where these areas are located.
Three regions correspond to the core of the distribution of C. estrellensis: the central Cerrado, the northeastern Caatinga and the centre-south. The National Park of Chapada Diamantina5 represents a sample of the thorny scrub vegetation of the Caatinga and fall within the distribution of the species. The National Parks of Brasília3, Araguaia2 and Emas6 are potentially important conservation areas in the Cerrado region. In the centre-south region, however, due to unrelenting pressure sustained since the 19th century on the original tropical/subtropical seasonal forest for agricultural land there are few extensive protected areas. There are, however, a number of smaller ones. There, Cariniana estrellensis is found in the National Parks of Serra do Cipó11 and Itatiaia7, the Ecological Station of Tupinambás14, the Biological Reserve of Poço das Antas15, the State Park of Rio Doce17 and the Forest Reserve Linhares20.
There is need for enhanced protection in a number of areas throughout the range of the taxon (cf Fig. 5).
| Table 5: | Type of information required for conservation strategy with current data of C. estrellensis |
 | |
 | |
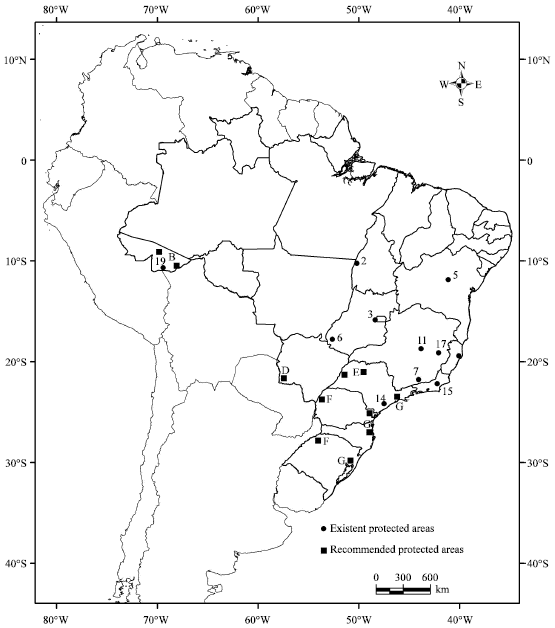 | |
| Fig. 5: | Distribution of existent protected areas: The National Park of Chapada Diamantina (5) in the Caatinga; the National Parks of Araguaia (2), Brasília (3) and Emas (6) in the Cerrado and in the centre-south region, the National Parks of Itatiaia (7) and Serra do Cipó (11), the Ecological Station of Tupinambás (14), the Biological Reserve of Poço das Antas (15), the State Park of Rio Doce (17) and the Forest Reserve Linhares (20) and the Extractive Reserve Chico Mendes (19), in Amazonia. Recommended potential protected areas in Brazil regarding Cariniana estrellensis conservation: in Amazonia at various localities in the State of Acre (B); in the centre-south (border between Brazil and Paraguay (D), interior of State of São Paulo (E), Araucaria forest in the states of Paraná and Rio Grande do Sul (F) and Atlantic coast forest in the states of São Paulo, Paraná, Santa Catarina and Rio Grande do Sul (G). Current protected areas based on IBGE’s (1993)map |
Even though it is reported present in the Extractive Reserve Chico Mendes19, the -species is at risk in Amazonia at various localities in the State of AcreB and in the centre-south border between Brazil and ParaguayD, interior of State of São PauloE, Araucaria forest in the states of Paraná and Rio Grande do SulF and Atlantic coast forest in the states of São Paulo, Paraná, Santa Catarina and Rio Grande do SulG. Protection of outlying populations is important due to the likely genetic variability existent in them when compared to the core of the range populations.
Basically, there are three practical options for tackling the conservation problem: traditional in situ reserves, normally in existing protected areas or specifically designated genetic conservation reserves; traditional ex situ stands, mostly provenance and progeny - sometimes silvicultural - trials; a compromise approach which is based on enrichment action to favour the targeted species.
Considering the current situation of protected areas in Brazil, the geographical distribution patterns of the taxon and the little that is known of its population structure, some recommendations can be put forward for genetic conservation programmes. Cariniana estrellensis, a normal component of typical forest formations, at its centre-southern occurrences in both Araucaria forest and Atlantic forest, stress should be on in situ methods with any ex situ/compromise action limited to more degraded areas subject to major pressure for agricultural land in the interior of the State of São Paulo. Such management action for compromise and ex situ approaches have to involve first of all firm commitment to safeguard from encroachment the areas chosen, particularly because they will be mostly in areas of intense agricultural activity in the centre-south. With present capabilities, Cariniana estrellensis is refractory in schemes of enrichment plantings and planted stands, possessing seed not amenable to nursery work because of poor germination levels and quick loss of viability. Nor is it suited for pure plantings-enrichment stands are more appropriate for it. Provision to exclude fire and protect seedlings recently planted out is vital - implying its use within established mixed stands.
With so many gaps in the silvicultural/management/conservation picture it is important that new initiatives should be on a planned and focused basis centred on identified priorities and not merely opportunistic. Characterising (and thereafter monitoring) population structure is a vital and immediate need. Institution of monitoring in natural populations could - and should - be associated with programmes of observation to elucidate the reproductive biology and breeding system more comprehensively.
The organizations with a mandate to advise on national conservation policy need to make a convincing case to the legal authorities and decision-makers of the implications of neglecting to declare the additional protected areas needed to secure a viable gene pool of the resources considered in this study and pinpointed in Fig. 5. One vital area of experience is in seed collection expeditions: already there has been successful collecting of other target species seed and, additionally, extending activities to storage and testing and nursery establishment would constitute logical extension of the work done on those species to others requiring similar action. Longer-term, more involvement in provenance and progeny trials - as has already started with other species - can be foreseen for other taxa. Teams dealing with reconnaissance surveys or inventories to identify areas (protected or with potential) containing the target taxon should also pay special attention to assessing population structure aiming at future rehabilitation (compromise) action. A major role is played by the ‘Instituto Florestal’ (Forestry Institute) of the State of São Paulo in forest genetic conservation in Brazil. Because most of the actions utilizing ex situ and the compromise (enrichment) approaches are concentrated in a region around that State, it is wise to have their collaboration in these programmes. In the south of the country, in situ conservation of Cariniana estrellensis in Araucaria and Atlantic forests could well be executed by researchers from Embrapa’s Forestry Research National Centre (CNPF), in Curitiba.
It is clear, after what has been discussed in this study, that despite the dearth of current knowledge, technical solutions for the conservation of forest genetic resources followed by action (breaking away the notorious verbosity generally encountered in the subject) are possible. Hopefully, scientific advice will be followed by political will to fulfil expectations.
The background level of effectiveness and versatility of the system of protected areas in Brazil is still poor, as far as conservation of forest genetic resources is concerned. Few examples of the main categories of protected areas with a bearing on conservation and management (national forest and extractive reserve) of the resource, through intervention, contain the species (cf Fig. 5). Indeed, the majority of existing areas containing Cariniana estrellensis are ‘indirect use’ categories (national park, biological reserve, ecological station), as shown in Figure 5 and no management actions are expected to favour the species studied here due to the non-intervention policy followed by Ibama. Such management actions are better directed to the conservation problem, in genetic reserves (in situ conservation) and formal planted stands (ex situ conservation) established by the scientific authority, as can be seen in Fig. 5. There is no initiative concentrating on alternative conservation approaches, such as enrichment planting, to complement traditional ones.
There has also been little implementation in the country of conservation options developed over the last few years from the increasing ecological understanding of tropical forest ecosystems. Much current activity takes insufficient account of the high diversity and low population densities of neotropical forest tree species. The vulnerability of the Cerrado formations to conversion for other land use makes application of effective conservation an urgent need.
Cariniana estrellensis owes much of its present knowledge to the late nineties’ generic revision of Cariniana for Flora Neotropica and ensuing wider interest and more research on the genus. Still, artificial regeneration and growth monitoring activities are receiving too little attention from the forestry sector. Apparently, the gap existent in the distribution range between occurrences in the Brazilian state of Acre and several locations in Bolivia and Peru and occurrences in central Brazil is due to poor collection rates in former parts of the species range.
Vital aspects important for traditional in situ and ex situ conservation are lacking and, additionally, there is poor legal protection in established areas. Compromise measures, basically through enrichment (eg mixture) with local seed source, are therefore urged to compensate for this.
As a conservation target, the taxon has limited background knowledge and future reappraisals may note important differences.
The following recommendations are made, with respect to the conclusions above, to assist the national conservation programme.
The patchy characteristics of forest formations in central Brazil Cerrado and the increasing fragmentation of more populated areas in the northeast and the centre-south, require that tackling the conservation problem should take into account the island-like nature of the habitats - habitats where conditions favour high rates of inbreeding, progressive loss of genetic diversity and, in the long run, extinction of species. To curb this, a network of local or, ideally, regional protected areas has to be established containing preferably ‘direct’ categories, but not disregarding the possibility of creating indirect categories when the opportunity arises. A significant increase in the number of even small protected areas contributing to a larger network would increase chances of survival of species for long term purposes. This network has to be supported by complementary approaches to favour thinly dispersed forest species by including sites which serve as links between more widely separated areas. This could involve the use of enrichment techniques with germplasm from nearby sources.
Cariniana estrellensis must be covered particularly by in situ conservation areas in the Cerrado and in the Atlantic forest, from where seed should be procured for expansion of artificial regeneration and growth monitoring activities.